Turquoise Energy News
Report
#178
Covering March
2023 (Posted April 8th 2023)
Lawnhill BC Canada - by Craig Carmichael
(CraigXC at Post dot com)
www.TurquoiseEnergy.com = www.ElectricCaik.com
= www.ElectricHubcap.com
Special Feature:
Federal,
Provincial
and Municipal Local Volunteer Grievance Panels: No
cost, timely and fair adjudication of minor disputes between
Individuals and Government (See in "In Passing")
Month In "Brief"
(Project Summaries etc.)
- Magnetic Variable Torque Converter - Renewable Energy Symposium
#2 - Magnetic Cooling with Gadolinium? Nah! - Copper Heatsinks? - A
Better Air Compressor? - A Working Battery! (for a while)... Exploring
Battery
Making Video Series - Solar Frame Fix - Magnetic Variable
Torque Converter - "New Chemistry" Battery Research & Development -
A Second Means of Taming Zinc Ions! - Exploring Battery Making Videos -
Plastic Recycling 2.0 - EV from Victoria Visit - Gardening
In Passing
(Miscellaneous topics, editorial comments & opinionated rants)
- Federal, Provincial & Municipal Local Grievance Panels
- Scattered Thots - ESD
- Detailed
Project Reports -
Electric Transport - Electric Hubcap Motor Systems
* Magnetic Variable Torque Converter with Planetary Gear: The Future of
the Automotive Industry!
Other "Green" & Electric
Equipment Projects
* Open Loop Air Heat Pumping
Electricity Storage:
Batteries
* This month's progress
Electricity Generation
* My Solar Power System:
- The Usual Latest Daily/Monthly Solar Production log et
cetera - Monthly/Annual Summaries, Estimates, Notes
This month has a special
feature outside the realm of 'green energy' and electric transport. I'm
relating what I believe should be a big help for people who seem to be
getting treated unfairly by government or big faceless corporations -
especially telecom companies - often by some part of a bureaucracy
applying bylaws and statutes in an unreasonable manner, making
unreasonable charges or providing poor service. A volunteer Grievance
Panel in each town or region, similar to a municipal Board of
Variance but broader in scope, gives people somewhere to turn when
they feel they are being or have been treated unfairly.
I have been thinking about this for some time, and have
finally written it up in semi-coherent form in the In Passing
section, below. I didn't get it finished until April... and then came
up with a couple of valuable modifications, broadening the scope to
include any "major institution" rather than just government
bureaucracy, even after I had initially posted this report. So I
rewrote it and reposted it a couple more times, and wasn't satisfied it
was good enough until April 10th. The changes are worth it! I will be
sending a paper copy to my member of parliament, who is someone that
just might like the idea and promote it energeticly.
Magnetic Variable Torque Converter
I tried for much of the month to get a piece of pure alume
for the rotor, to get more electromagnetic interaction with the magnet
rotor than with alloy. Double might be about right. I knew it wasn't
common, but I wasn't prepared for this... Nobody seemed to have it. at
all. One place said they could get it in but I'd have to buy a whole
sheet. That would be well over 1000$ - plus shipping! Finally one place
said they would sell me a piece, 1/4 inch thick instead of 1/2 inch, 2
x 2 feet... for 1100$! Yow!
At that point, I decided copper would be (much) cheaper,
available, and after all the best - the most conductive - more than
double the alume alloy, so I could use a thinner piece. Then someone
had many Kg of copper bits and I bought them from him at copper spot
price. So instead of buying a slab I started thinking I would cast the
rotor in copper. A concern is that the crucible only holds 1Kg of bits,
where the rotor will need about 2-1/2 Kg of copper. Will it work okay
if the copper is cast in two or three melts to fill the mold instead of
all at once? Somebody on youtube said you shouldn't do that. I guess
I'll find out. But first I'll try casting copper heatsinks for a
Peltier cooler for practice, and also to see how much better the cooler
performs than with alume alloy.
Another guy on youtube said he made crucibles out of fire
extinguishers. I could perhaps make one large enough for 3Kg of copper
out of a propane bottle. (carefully purged before cutting!) That
wouldn't fit in my little metal furnace, but perhaps I could fit it
into my mini-kiln. OTOH, he was just melting alume [alloy, sigh!], a
lower temperature, and he said they only lasted a few pours. Would it
last one pour with copper?
This is sure some serious "scope creep" just to get a
metal disk I thought I could simply buy!
 There was a
second Renewable Energy Symposium on the 11th &
12th, at the Haida Heritage Museum, following the one in September 2018
in Old Masset centre. I decided to get a table and show mainly the
battery and torque converter work I had been doing, plus the updated
handheld bandsaw mill and some "Plastic Recycling 2.0" molds and
moldings.
There was a
second Renewable Energy Symposium on the 11th &
12th, at the Haida Heritage Museum, following the one in September 2018
in Old Masset centre. I decided to get a table and show mainly the
battery and torque converter work I had been doing, plus the updated
handheld bandsaw mill and some "Plastic Recycling 2.0" molds and
moldings.
I said I was interested in "TLL Yhada" and the 2 MW solar
project at Masset Airport. The panels will cover the grassy field to
the side of the runway, and batteries will provide power at night.
The previous HDPE
melts had had no discernible smell in the kitchen oven, but the odor
with the PP
ropes that started out with a big air space between the mold's lid and
the wall was quite objectionable.
 I did a few
new "sample size" pieces in the kitchen oven the week before to take to
the event, where I discovered, since I could control the temperature,
that 400°F was about right for both PE and PP.) I started typing up
information pages about the battery research on the 5th, even before
finishing the February TE News Report.
I did a few
new "sample size" pieces in the kitchen oven the week before to take to
the event, where I discovered, since I could control the temperature,
that 400°F was about right for both PE and PP.) I started typing up
information pages about the battery research on the 5th, even before
finishing the February TE News Report.
BTW - Smell of Melting Plastic: It would seem from this limited
experience that melting polyethylene has little or no odor, but
polypropylene smelled pretty strongly.
I will not hesitate to do even a big piece of HDPE or UHMW
in the oven in the kitchen next time, but an outdoor oven seems to be
needed for polypropylene.
The weekend went by quickly with much talking to
interested people. I said to those that stopped, "Here it is: four
totally different projects." There were people interested in each of
them.
Mid morning of the 15th
("afternoon" according to the clocks, which for no apparent reason and
seemingly against the wishes of much the general population have gone
ahead a second hour onto "afternoon wasting time"): Yet another dusting
of snow last night, and now it's again pouring rain or sleet, 3.6°
outside my door. It's been like this daily since mid to late February,
except sometimes with strong wind too. Occasionally there's a bright
spot in the clouds where the sun is. I'm getting tired of it, and it's
keeping all outdoor projects at bay.
Magnetic Cooling with Gadolinium? Nah!
I saw a youtube video about air conditioning. Something
stressed as a "horrible problem" was the amount of electricity they
use, putting a strain on power grids. Somebody has to explain to me why
air conditioners wouldn't be powered by solar panels. Opposite of
heating where there's no solar power when heat is needed, the hotter it
is the more likely there's plenty of solar power to run an air
conditioner.
Another thing in this video was a brief mention of "other
ways" besides compressor/refrigerant cooling, including magnetic
refrigeration. That of course would use gadolinium. That got me
thinking again that there are surely means of heat transfer to and from
the gadolinium by solid copper (or graphite) instead of the complex
(and may I say bizarre?) ways of doing it with helium gas and the like.
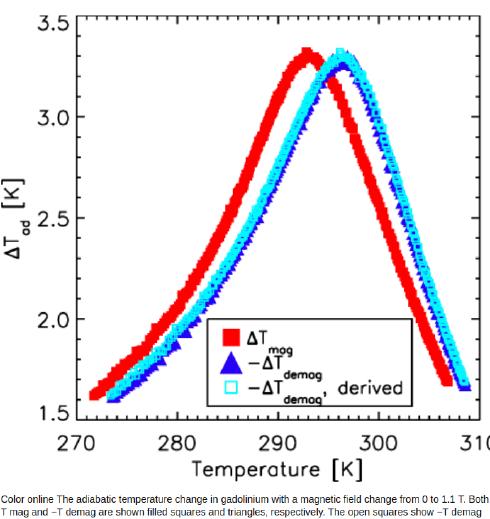
A graph shows temperature change in gadolinium entering or leaving a
1.1 Tesla magnetic field being about 2 to 3.5°C or °K, maximum effect
around 295°K (22°C) and down to 1.6° change at freezing. (Gadolinium
alloys can modify the center temperature, so maybe 3° everywhere in a
fridge or freezer, so maybe 8 stages to allow 24° fridge cooling?) One
Tesla is "neo magnet" permanent magnet territory. Or maybe the field
can be got up to 2 or 3 Teslas (Hallbach magnet configuration?) and
need fewer cooling stages.
 I visualize a
piece of gadolinium coated on opposite sides with copper or graphite,
perhaps moving back and forth. On its outward swing it contacts a
graphite coated magnet, which is cooled by a fan. So it heats up, but
its heat is drawn through the copper and or graphite. Then it goes the
other way where it cools as it exits the magnetic field. Here the other
face (or better, somehow the same face) contacts the cooling side
copper and transfers its coolness into that. If instead of a back and
forth "swing" it is traveling say in a circle, the copper/graphite heat
transfer contacts could be rollers - or the Gd could be in the form of
a roller - to avoid friction. Each stage transfers to the next by a
solid copper or graphite bar, hot to cold, hot to cold.
I visualize a
piece of gadolinium coated on opposite sides with copper or graphite,
perhaps moving back and forth. On its outward swing it contacts a
graphite coated magnet, which is cooled by a fan. So it heats up, but
its heat is drawn through the copper and or graphite. Then it goes the
other way where it cools as it exits the magnetic field. Here the other
face (or better, somehow the same face) contacts the cooling side
copper and transfers its coolness into that. If instead of a back and
forth "swing" it is traveling say in a circle, the copper/graphite heat
transfer contacts could be rollers - or the Gd could be in the form of
a roller - to avoid friction. Each stage transfers to the next by a
solid copper or graphite bar, hot to cold, hot to cold.
But I'm still waiting... since about 2015 now... for a
breakthrough Peltier module with a higher COP at higher temperature
differentials. That would probably make the whole magnetic cooling idea
obsolete and not worth pursuing. I think it will happen.
Somebody was recently claiming a room temperature
superconductor (seen on WION, youtube -- I heard of one in the 1980's
or 1990's, too! The new one sounded like a quite different alloy.) and
I'm sure various such "groundbreaking" things are inevitable given
enough time.
Copper Heatsinks?
I did have one further thought: If I have to cast a copper
rotor for the torque converter, maybe I should try casting something
smaller and simpler first? How about sand casting a copper heatsink, in
a simple, open mold? In fact, having recently charted the performance
of the Coleman camping cooler with its alume heatsinks (TE News $174), I could
take impressions of those in sand, fins down, and cast them, then see
how much colder the cooler would get. Shouldn't need air vent holes
that way. I'll have to see how flat or bulging the top flat face comes
out. It has to be honed to a perfect flat.
It would be interesting to see how those perform in the
cooler. (If they do as well as I hope, I'll do a big one for my Peltier
module shallow chest fridge!)
A Better Air Compressor?
 Moving right
along... also on youtube I saw a rather radicly new design of air
compressor, the "Lontra", much more efficient than any previous one. It
had two rotors at 90° to each other. Through most of the cycle the main
rotor acted like a circular bicycle pump, pushing air out the outlet
port. Then it reach the "end" just past the output port, and the gap in
the other rotor, spinning synchronously, reached the gap in the main
one and they passed through each other, virtually but not quite
touching, letting almost no air pass between and closing off the output
port. Meanwhile the whole circle behind the main "piston" had filled
with new air to be compressed on the next cycle. With narrow air gaps
covered by wide pieces for low leakage there were no parts in friction
with each other, and motion was continuous rotation rather than
reciprocating.
Moving right
along... also on youtube I saw a rather radicly new design of air
compressor, the "Lontra", much more efficient than any previous one. It
had two rotors at 90° to each other. Through most of the cycle the main
rotor acted like a circular bicycle pump, pushing air out the outlet
port. Then it reach the "end" just past the output port, and the gap in
the other rotor, spinning synchronously, reached the gap in the main
one and they passed through each other, virtually but not quite
touching, letting almost no air pass between and closing off the output
port. Meanwhile the whole circle behind the main "piston" had filled
with new air to be compressed on the next cycle. With narrow air gaps
covered by wide pieces for low leakage there were no parts in friction
with each other, and motion was continuous rotation rather than
reciprocating.
 The wide "seal" coverage in a
small area - a tiny air gap with no parts touching - has much
The wide "seal" coverage in a
small area - a tiny air gap with no parts touching - has much
less leakage than a "screw compressor's" large area seals with narrow
coverings of the gaps.
This might be just what is
needed for high performance with the open loop air heat pumping.
Apparently a company with a number of large compressors was saving 30%
on electricity for them by switching to these. That's huge! That's as
much saving as recovering the remaining compression after extracting
the heat from the compressed air. (If it could use the remaining
compression of the cooled air too, perhaps with a separate piston ring,
the COP could go up, up! If COP 10 sounded unrealistic before, it's
definitely getting closer with these!)
Sounds like it's just as well I never got around to
building the tricky "ROVACS" or the complex "Liquid Piston" rotary
design compressor yet! This would have to be simpler to make, and I
can't help but think there must be a way of making a small "Lontra"
type with just one rotor and some air closing flaps, and some even
simpler end-of-stroke "move away to let the piston past" mechanism.
(Hah! - Have a steel piston moved along by magnets in an enclosed
non-magnetic tube needing no leaky openings to connect it to the motor?
And the magnets also move/activate the "end-of-stroke" mechanism? And
if the pressure is too high, the magnets can't hold the piston and let
it slip, preventing dangerous pressure buildup if something is blocked?
Hmmmm! Fantastic idea... or just Silly?)
OTOH, for a small, low power compressor, why wouldn't an
actual reciprocating "bicycle pump" mechanism with low inertial mass be
just as good, or close enough? It would certainly be simpler to make
than any "Lontra knock-off" I can think of making. It only needs to go
up to 50 or 60 PSI for the heat pumping application. (Most compressors
shut off around 120 PSI.) The lower pressure should relax the
requirement for robustness considerably.
A Working Battery! (For a while)
I started the month by trying manganese-zinc with all the
new techniques. I was hoping that the acetone treatment would make the
Mn2O3 / MnOOH (valence III) recharge to MnO2 (valence 4), but it didn't
work. Performance continually deteriorated.
So I took out the MnO2 side
and put in a nickel oxide (NiO) mix also containing my usual monel
(cupro-nickel ~65:35), samarium oxide and calcium oxide. With all the
latest refinements. With a [leaking] wax seal. This did work. As usual
starting apparently totally discharged, the performance gradually
increased over two days and more, and it would put out 4 or 5 amps. A
for real, working battery!
How ironic to start out with the idea that there must be
battery chemistries that haven't been tried, and end up with common
nickel-zinc as the first combo that's working well. But even in that
there are things in that that are new and unique. Nobody seems to have
done it before with KCl salt electrolyte, and no one else seems to have
discovered that unlike all single element metals, cupro-nickel doesn't
oxidize away in the positive electrode at moderate alkaline pH levels.
Somehow the copper and nickel seem to protect each other.
Hopefully the nickel powder mix can't penetrate the
methyl-benzene treated paper and the zinc ions can't penetrate the
sodium dodecylbenzenesulfonate painted into the same paper and so
nothing will start making low resistance paths between electrodes.
Solving the zinc ion problem (as I've said before) has been a "holy
grail" of battery making for 150 years - including another 15 since I
began my own experiments.
The other thing that has revolutionized my previous
generally poor results is soaking the positive electrode powder mix in
acetone (methyl-methyl keytone). I can't say I understand it clearly,
but I think it dissolves the oxides and makes for epitaxial crystal
growths, mixing different substances at a more molecular level instead
of just separate jumbled powder particles. The difference is a couple
of orders of magnitude in current capacity.
But the cell leaked. I kept filling it, but after a while
performance started deteriorating. Thinking about it, the SDBS in the
separator sheet might become more and more diluted among other
problems. pH would start to drop if the Ca(OH)2 got too diluted, too. I
think I'll go back to the idea of potting the cells in epoxy to prevent
any chance of them leaking.
 Toward the end
of the month I turned the cell design sideways and put some tiny holes
in, to let gas bubbles rise over a short path and into the upper
reservoir, which can hold more liquid.
Toward the end
of the month I turned the cell design sideways and put some tiny holes
in, to let gas bubbles rise over a short path and into the upper
reservoir, which can hold more liquid.
 I started
making a new cell (3D printed front cover) but didn't get it finished
owing to competing attractions. (Just needs compacting + epoxy, and a
new shape of alume clamps.)
I started
making a new cell (3D printed front cover) but didn't get it finished
owing to competing attractions. (Just needs compacting + epoxy, and a
new shape of alume clamps.)
Another Means of Taming Zinc Ions!
Assuming indeed using SDBS stops zinc ions as it seems to
in my experiments so far and hence makes a zinc cell last far, far
longer, there now seem to be two ways of doing it, because someone else
claims to have prevented zinc ion migration with modified chitosan
(crab shell material). I plan to look at this further as their
technique of using it was different than mine of painting it into the
separator sheet. They impregnated their material with zinc (powder?)
and then put it right on the surface of the zinc of the electrode.
Exploring Battery Making Videos
Regardless of success or lack thereof, I have accumulated
a number of exciting developments/discoveries worth sharing and I
started my planned Battery Making video series on Youtube with four
videos:
Exploring Battery Making with Craig - Intro (Long - 25
minutes)
https://youtu.be/R0_ZkBFefDk
Exploring Battery Making #2 - Preparing a Separator Sheet
https://youtu.be/8-MaCfFwvbY
In this I've given away two of my greatest "secrets":
about the toluene for preventing penetration of the paper by
nano-powders, and about the SDBS which I believe is the answer to the
150 year quest for trying to keep zinc ions from killing batteries.
Exploring Battery Making #3 - Getting/Making Sodium
dodecylbenzenesulfonate
https://youtu.be/F_7Vj8SDw0w
This one is about extracting SDBS from Lemon Fresh
Sunlight dishsoap by ersatz "chromatography" in case others find it
as hard to buy as I did.
Exploring Battery Making #4 - A Zinc Electrode
https://youtu.be/JjwtgkQnwzM
I had also earlier taken clips of making the powder for a
nickel manganates electrode. I put them together and added another one
at the end about dissolving the powders in acetone to get everything
mixed at the molecular level within epitaxial crystals. So I have the
video. But I haven't proven that I've made ~~NiMn2O4 yet, or that it
works as a rechargeable electrode.
Then, with the next cell all ready to close up and epoxy,
didn't get back to it in March. I want to make sure it actually works
before I publish the nickel manganates video, and to put up the actual
electrode assembly video and at least initial testing of the cell at
the same time. But somehow it's "work outside" season again and the
days have too few hours.
Solar Frame Fix
 In February a
powerful wind with even more powerful gusts blew over my "A" frame of
solar panels on the carport roof. These three panels installed in this
sunny location out of the tree shadows at a good angle (45°) last May
(2022) add more output to the system than any other three panels. After
waiting for better weather I finally got the frame repaired and screwed
back up on the 22nd and 23rd. One panel was broken so I set it up with
two. (I don't think I could have lifted it with all three panels on it!)
In February a
powerful wind with even more powerful gusts blew over my "A" frame of
solar panels on the carport roof. These three panels installed in this
sunny location out of the tree shadows at a good angle (45°) last May
(2022) add more output to the system than any other three panels. After
waiting for better weather I finally got the frame repaired and screwed
back up on the 22nd and 23rd. One panel was broken so I set it up with
two. (I don't think I could have lifted it with all three panels on it!)
Then it got windy so I stayed off the roof, and then I cut
down three alder trees. The biggest one was leaning and in spite of a
rope on it didn't fall in the direction I cut the hinge. It hit my
neighbor's tractor shop and bent the gutter. (thankfully not worse! and
thankfully he's not around at the moment.) I had to repair that and get
the limbs and tree off his property, so other things got put off.
But after well over a month
since the wind incident I couldn't bear to start April with one pane
still missing, so I went up on the morning of March 32nd, got a new one
up on the roof without dropping it on the way up, bolted it on and
reconnected the wires. I soon saw 850 watts on the power monitor from
those three panels plus the two on the pole. It started out a nice day,
but a couple of hours later it was snowing!
 The 2 solar panes on the pole and
the 3 on the carport roof are quite visible from the highway...
The 2 solar panes on the pole and
the 3 on the carport roof are quite visible from the highway...
so if nothing else it's bad publicity to have them broken or missing!
I must confess that I had
been careless in the original (and in fact never quite finished)
installation. Some of the "no screws showing" roofing sheets on the
carport had slid down a bit, and I had wanted to reposition them before
putting in screws on a couple of legs of the frame, so a couple of the
six legs weren't fastened. And other screws only went through the sheet
metal roof into nothing. Those ripped out, then with the back free the
frame would have tipped forward in a blast of wind and the screws on
the front side were yanked out or broken. (I had just used 3 inch #10
deck screws, but I'm pretty sure they'd have held fine if all all the
legs had been secure.) This time I had adjusted the slidden roofing (as
easy as tapping the bottom edge with a hammer with a wooden block
between - if only I'd known) and I fastened pieces of wood in the
"ceiling" under the roofing to securely hold the screws of the three
back legs. It would take a substantially bigger hurricane to yank it
off now. (Then again, looking at some tornado-destroyed towns in
southern USA in March, anything is possible these days.)
EV from Victoria Visit
 Someone named
Angus showed up here on the 9th in an old Subaru Brat 4 wheel
drive pickup truck he had converted to electric. It had a
Nissan Leaf motor and three big Tesla car batteries behind the seat.
(36KWH? or was it more?) I forget what the motor controller was (a
Tesla model, IIRC), but there wasn't room under the hood for the
regular Leaf controller, and he had got up to 320KW out of the motor
that the Nissan controller normally limits to 80KW!
Someone named
Angus showed up here on the 9th in an old Subaru Brat 4 wheel
drive pickup truck he had converted to electric. It had a
Nissan Leaf motor and three big Tesla car batteries behind the seat.
(36KWH? or was it more?) I forget what the motor controller was (a
Tesla model, IIRC), but there wasn't room under the hood for the
regular Leaf controller, and he had got up to 320KW out of the motor
that the Nissan controller normally limits to 80KW!
It seems there are now enough EV's at auto wrecking yards
that one can do some lovely economical EV conversions if one can figure
out the microcontroller stuff that commercial EV's are full of for the
batteries and controllers. He got the truck in Mexico and so it was in
good condition instead of rusted out.
 He had come up from Victoria to
work tree planting, and someone there (Tom!) said he should stop in and
see me.
He had come up from Victoria to
work tree planting, and someone there (Tom!) said he should stop in and
see me.
He had 3D printed a J1772 fitting. He charged his truck at
my charging station and we talked about EV's and such for a couple of
hours.
He had also charged at a new charging station in Masset.
Fine thing - I've been waiting for them to put one in there for years
and I have to find out it's been done from some visitor to the island!
He had designed some coupler plates and other fittings to
fit the Leaf motor to his transmission et al, and found there was a
market for them, so he started BratIndustries.net and now sells them
for several vehicle models.
He had driven it to Rose Spit along the beach at low tide.
Yup, you want a four wheel drive to do that. That beach is notorious
for claiming vehicles that get stuck in the sand until the tide comes
in!
Gardening

Well, it was too cold to do
much outside. Even into April it was pretty chilly. (and in 3 weeks the
blackflies will be out!) I was too busy to get any seedlings going.
(Ag, I'm late!) Over the winter the cherry tomato and peppers in the
bay window with a light over it continued producing fruit. I had put up
'tablecloth' curtains to reflect the light back at the plants, but that
made it pretty dreary inside and I mostly unhooked them. The lettuce
under the LED light also kept making leaves, which I plucked as desired.
Vainly hoping it just might come back in the spring, I
kept watering the walnut tree daily all winter. (If only I had done
that when it had leaves!) No sign of anything so far.
I put down some cardboard from grocery boxes to kill the
grass for gardning. The idea was great. But from last time I did it 3
years ago, I'm still plucking bits of packaging tape from the garden,
so this time I decided to pull it off the boxes in advance. It's a
party killer! 2-1/2 hours of peeling tape off the cardboard, and then
ten minutes to lay them out on the ground. The fact of packaging tape
stuck on the boxes magnified the time I spent doing it tenfold!
 Overgrazing
makes deserts. Here, although it's too damp for a desert, my own
chickens illustrated overgrazing when I moved their yard over a little
bit. The green strip didn't last.
Overgrazing
makes deserts. Here, although it's too damp for a desert, my own
chickens illustrated overgrazing when I moved their yard over a little
bit. The green strip didn't last.
I'm making a smaller yard, with wheels, that will be far
easier to move to new sections of lawn. This large, pivoting-corners
one was supposed to be able to move easily, one wall at a time, but
when I had to staple chicken wire over the roof to keep critters out,
it didn't really work. (And chicken wire isn't good enough anyway.
Raccoons can rip it off or tear it. I'm using heavier fencing wire on
the new one.)
In
Passing
(Miscellaneous topics, editorial comments & opinionated rants)
Grievance
Panels
of Local Volunteer Citizens
to Hear and Rule On
Individual Complaints about Dealings with Major Organizations
(Concept Outline)
PREAMBLE
In a democracy ultimate power of decision rests with the
people. Individual leaders come and go. Autocracy in any form cannot be
sustained over a long term. Only representative democratic government
can be made socially sustainable over the generations, and only if its
enduring institutions are structured so that they can evolve and solve
each underlying problem that arises in ways that benefit society, as
that society grows and changes over the years, decades and generations.
With the gradual rise of large, bureaucratic institutional
organizations, new structures of governance have been needed for long
over a century to bring effective representative/democratic government
back on course. The problem is that until a new structure has been
invented, until a new concept is brought to light, we don't know what
it might be and we are groping in the dark to "put out fires" because
we have not created a structure for solving the underlying problems
that cause the fires.
Here is an idea that I believe could go a long way toward
restoring fair and equitable treatment of society and individuals by
institutions and the bureaucracies that have grown up around them. It
is surely only one of several new things that need to be created to
obtain social stability and eventually social sustainability, but I
think it fills in one existing "democracy power vacuum" blank in the
picture quite well.
Major institutions or organizations have over many decades
built bureaucracies which are faceless and anonymous. Examples of
"Major Institutions" include government at all levels local through
national, corporations especially such as utilities, and educational
and health institutions.
When members of the public have a problem with one, they
are generally unable to meet or to be heard, directly or indirectly, by
those heads of such institutions who have the decision making power -
only with employees who are not empowered to solve or smooth over many
of the problems an individual or the public in general may have in
their relations with that institution. Many examples could be cited. (A
few follow down below.)
As a result, our major institutions have become feral.
They are no longer bound to be and too often are not, tame, civilized,
responsive and reasonable in their dealings with the people and society
they exist to serve.
Sometimes an employee may even be part of the problem.
Civil enforcement officials may apply bylaws or terms in situations
which were not envisioned by legislators, and the results work harm
instead of good, often to individuals, families or small business
owners caught by unreasonable demands.
What recourse do people have? They can hire a lawyer and
go to an overworked court of law, but the verdict may not come soon
enough to matter, and even after all the expense and time taken from
their productive work -- which may generally cost them more than their
budget can bear and more time than they have, the judge might have to
rule that, yes, such is the law and it has to be applied equally and
complied with regardless of its appropriateness or fairness in this
individual situation.
So with "the cure being worse than the disease" (and
perhaps ineffective too) people usually simply submit to what they feel
is an injustice, undesired by society as a whole and unintended by
their elected representatives but demanded by bureaucracy.
The Grievance Panel provides a local place
for the Complainant to be heard in good time by his
respected peers to gain what they collectively, on the facts presented,
consider to be a fair decision or settlement in his dealings with any Major
Institution without the lengthy and costly process of filing
suit in a formal court of law, and unlike in a court regardless of
whether a demand being made is technically legal or not, and
without reference to "precedents" of similar but rarely identical
situations. Panel judgments based on interpretations of reasonable
versus unreasonable, fair versus unfair, will
help prevent misapplication or abuse of authority and of corporate
power. Repeated complaints on a specific issue or directed at a
specific institution will help point out problem areas.
A local Grievance Panel would be similar to the present municipal
Board
of
Variance, but of broader scope, operating in each locality
on complainants' issues regarding any major institution, public or
corporate. It is composed of peers - volunteer citizens not in the pay
of the government or the corporation involved, who have not contributed
to the antecedents of the situation and who have no personal stake in
the outcome.
While most complaints about government are likely to
relate to property, and most complaints about utility corporations to
unfair charges or poor service, the scope of the panel is not to be
limited to those areas.
All panel decisions may be appealed to a court of law if
either party is dissatisfied with the ruling. The onus is on the losing
party to initiate legal action if desired. Rulings in favor of the
complainant may require the government or a corporation to cancel a fee
or fine levied or to promptly refund money already paid, to cease
restrictive requirements, to permit actions it was prohibiting, or to
provide a service it was withholding.
In the event that panel members are really unsure how to
rule, if the case is too complex or vague, they may elect to not make a
ruling. A tie vote is also considered to be "no ruling". The ruling (or
lack thereof) will be written down and signed by the panel members, and
both parties will receive a signed copy on paper.
Here are some suggested parameters. These have not been
thoroughly thought out or researched, and so further consideration
before actual implementation is desirable.
1. These local panels give individuals a place to turn for a speedy
decision if they believe that a major institution or an agency or agent
of that institution has demanded something unreasonable, unfair or
unjust from them, or is refusing to provide something it should. It
hears only civil matters, not criminal, involving only disputes between
any major institution and a citizen, household
or personal small business, and its decisions may be overruled
by any court. The onus is on the disagreeing party to take it to court
if it wishes to contest the decision. The scope of the panel is not
limited to any particular subject or area.
2. This is a volunteer advisory panel comprised of one's peers,
concerned local citizens. It is anticipated that many or most civil
disputes will be settled agreeably at this local level without recourse
to law courts. Preferably the panels will come to have some experienced
and respected people as well as a few younger students or beginners.
Judgment should always be a group function to reduce the effect of
individual biases and there must be more than two persons on a panel.
(I'm anticipating three will be the most popular number - after all
they are volunteering their time - but four or five or even more is
certainly possible, especially if new members are in training.)
3. An individual who believes a demand or problem related to an
institution is unfairly harming his interests may apply to be heard by
the panel by any convenient means. This complainant (or applicant)
will
be
heard personally, and no monetary charge will be levied for the
hearing.
If, for example, a fine, fee,
action demand or refusal to act itself was asked by the institution,
the institutional party will be asked if they wish to present their
case, in writing or at the hearing or both. Preferably the applicant
should also make his case in writing before being heard live. A form
for doing so may be provided. "In writing" may include submission on
line. In addition to releasing the complainant from a demand, or
demanding that the institution act to provide service to the
complainant, the panel in finding for an applicant may rule that fees
or penalties pending or applied should be promptly canceled or refunded.
4. The panel will meet regularly not less than monthly (or more often
as is deemed desirable) at scheduled times posted in advance, in an
agreed public meeting space provided free in a government building. If
there is insufficient time to hear all cases, another meeting shall be
held within 2 weeks to clear the backlog, and every two weeks
thereafter if and as necessary. If there is more than rarely too much
work to render speedy judgments for all, a second panel of further
volunteers may be required even in the same locale.
5. Selection of Grievance Panel members. Any citizen may volunteer or
may nominate someone to be a member of the panel, and if seconded such
nomination shall stand for three years. I have no clear idea who should
select members from the volunteer list. Perhaps present panel members
should select them, or at least have a good say. Members of nearby
panels might also be called in for the decision. Terms will be for
(?)two years, and no one shall serve for more than two consecutive
terms. (ie, (?)four years.) Those with violent criminal records should
be excluded, and others with criminal records as well unless the seven
year statute of limitations has passed and they have committed no new
breaches. (Again they would still have to found acceptable by the other
members.) Municipal councils might perhaps have a veto power over known
"troublemakers" - the antisocial and those not of sound mind.
I anticipate that owing to the local, volunteer and
plentiful nature of grievance panels and their diverse fields of
activity, it should be very difficult to "stack" grievance panels in
any meaningful way to the benefit of any one major institutional entity
or related group of entities, but perhaps this aspect of panel member
selections should be thought through to pinpoint any possible
"loopholes".
6. For the purpose of eligable institutions, "major" should
be defined, or specific institutions should be denominated as "major"
and the list updated when required.
7. The volunteer panel members shall not be liable to penalties or fees
as a result of their decisions, including if a decision is overturned
by a court.
8. Doubtless parameters already exist for the operation of municipal
boards of variance, and one suspects they may differ by municipality,
in different provinces, or in different countries. At least a few of
these should be studied before going too much farther than the above on
setting up operating parameters for grievance panels.
Some Examples of Apparently Unfair or Unintended Situations
Potentially Warranting Complaint
1. (This happened to a friend of mine - his own parents' property after
they passed on. He tore down the old house and was going to build a new
one. And I'm pretty sure I've heard of similar cases, although
specifics aren't coming to mind. The municipal inspector made no effort
to help him solve the difficulties his requirements imposed. He had
nowhere to complain about these requirements.)
The homeowner can't get needed permits for months or even years. He has
nowhere to complain and finally sells the property as-is in
frustration, but the developer who buys it quickly has all the needed
approvals - surely because he knows how to "grease the wheels".
2. A BC couple decided to buy farm land in a rural area in Nanoose and
grow produce, and to make their own restaurant serving that produce on
the property. They looked into everything first and carefully planned
it, but when they actually went to do it one absurd hurdle after
another was set before them by local officials.
One I recall was that while they had not intended to serve
alcohol at all, but owing to a BC bylaw intended to protect BC local
wineries they were forced to get a liquor license, divert acreage to
growing grapes and hops and serve alcohol. If they weren't even going
to serve alcohol, their restaurant should have been completely outside
the scope of this bylaw.
There were others. The last one, having already seriously
diverted and delayed their plans and spent tens of thousands of extra
dollars to comply, was that they weren't permitted to build their long
planned house because they already had built the restaurant and weren't
permitted to have two buildings on one farm. [Huh?]
They were in a rural area where there was no Board of
Variance because rural regulations are supposed to be few and simple.
Any rational person on a grievance panel surely would have looked at
any of the demands and said that they were outrageous. (And if that got
the unreasonable employee(s) tamed or fired, so much the better for
everyone! I'm sure no elected officials had any idea all this was
happening and would have been aghast. But would they have been able to
fight their own bureaucracy and change it? A grievance panel would (I'm
certain) rule in favor of the enterprising couple.) I saw the story on
youtube.
3. I have heard that BC decided after some years of severe forest fires
to ban burning after March 31st. That may be reasonable in the south
where it's already starting to get a bit dry, but in the north the snow
hasn't even melted yet, and even here on the coast it was too cold to
work outside until the last few days of March. I'm cutting trees for
firewood into early April ASAP before there's too much sap. Now I'm
supposed to leave the branch piles on my lawn to get tinder dry, until
next winter? That's definitely more of a fire hazard than burning them
this spring after they dry enough to burn. But if I do might I get a
$1000 fine? In some areas burning might not be practical until May or
June. Perhaps not the concept but the specific dates applied to the
whole province need to be modified by region. (OTOH we already had laws
about burning and fire hazard ratings that depend on whether it's wet
or dry! Don't those make more sense? And aren't most forest fires
started by lightning?)
4. There was a news story (USA) where police shut down a childrens'
lemonade stand for "running a business without a business license". (I
think they may have fined the parents... can't remember. The whole
story sounded absurd.)
5. Someone (USA) had grown a vegetable garden in their fenced front
yard for years. A municipal employee came along and told them to rip it
up and plant lawn immediately or be fined 1000 $/day! (After hearing of
this the municipal council specifically made a new bylaw permitting
vegetable gardens in front yards, so what the employee demanded was
clearly not the intent of the city's bylaws! By then they had had to
rip out their garden.)
6. The Old Tobacco Shop (I probably have the name not quite
right), for ages in downtown Victoria, was told by a municipal employee
that advertising tobacco was illegal and that he would be fined if he
didn't take the store sign down. Another municipal employee said his
sign was antique and came under heritage designation, and that he would
be fined if he removed it.
Likewise, someone was told to remove a dam on his property
or be fined. (IIRC he actually wanted to remove it.) But the dam had
been built by beavers and the wildlife department told him he would be
penalized if he removed it.
7. Not only government but other large institutions sometimes need to
be reigned in. Feral Canadian telecommunications companies are
notorious. One fraud was perpetrated by Telus in (?)2009: when people
(fed up with high monthly charges) switched en masse to Shaw
phone/internet/cable TV service, which had recently solved technical
problems and become widely available. Telus pretended to not know and
continued billing people as if nothing had changed. They did it to me.
They lied to me twice on a phone call. Most people would fear for a
black mark on their credit rating and pay for some extra months. As an
electronics tech I had tested the line and knew they had cut it from
their office when I switched. A formal complaint to the "commissioner
of complaints for telecommunications services" ("CCTS") in Ottawa got my
fraudulent charges dropped - it didn't start a criminal fraud
investigation or help anybody else. As usual it wasn't worth any
individual's time to take Telus to court or start a class action
lawsuit.
Then in 2017 when I moved from Victoria to Haida Gwaii,
people simply couldn't get through to me. It turned out later that
Telus was deliberately and brazenly blocking calls to cell phones all
across Northern BC. The motive would seem to have been to force
everyone to switch their mobile phone service from "whoever" to Telus.
The orders for such outrages have to come from the head. No subordinate
would dare attempt to foist such schemes on the public.
After who knows how much trouble they caused to how many
people, the CRTC forced them to stop blocking calls. But instead of
arresting this CEO for Betrayal of Public Trust, in 2021 or so
a former head of Telus was appointed head of the CRTC - doubtless this
same criminal who had caused all the trouble in previous years! (He was
soon spotted in a pub having a beer with the CEO of Bell Telephones, a
company that the CRTC is supposed to be regulating.) What are the
chances now of getting positive action from the CRTC when there's a
problem with Bell, Telus or other major telecom companies?
Dozens of complaints upheld by independent grievance
panels across a region or nationally could not be hidden away in a
drawer at the CRTC and would in fact constitute abundant evidence of
failure of fairness by the institution or criminal conduct by its
leader.
Probably all of us can think of things that have been unfairly/unjustly
put upon people, even on themself. I should think an impartial place to
take such complaints to be heard by one's peers, those unpaid
volunteers who didn't help create the situation and who have no legal
obligation to say "Well, I'm afraid that's the law (or "the way it is")
so suck it!", is exactly what's needed to iron out many of the problems
individuals have with major institutions, to bring some fair decision
making power back to the people and, with local Grievance Panels
everywhere, to get timely decisions instead of too costly and too long
delayed.
An Opposite Example
Here's what can happen when there's an impartial volunteer
panel of one's peers to complain to:
1. Long ago (1978?) I met someone who took his complaint to a Municipal
Board of Variance. A corner of his garage stuck out a few inches and
the eaves at that corner maybe 18 inches too far onto the boulevard and
he was told by a city official to move or demolish the garage. It had
been that way since before he bought the place and was in no ones' way.
No one had complained about it. Technically the official was within the
law, and a court would probably have so ruled. It's easy for government
to put heavy burdens on individuals and families virtually on a whim
when there is no penalty to itself for doing so.
He said he went in with all kinds of documents, photos and
arguments, ready to fight. They looked at his application for a
variance for a moment, and probably the photographs, (or had already
looked at the submission?) and without further comment the head of the
board said "Granted." and it was signed. No need to tear down his
perfectly good garage!
Isn't that how it should be? My proposal is essentially to
extend the scope of such independent local tribunals, and allow them to
work with a complaint of any sort against any major institution.
Grievance panels everywhere may pass on unspecified minor matters of
contention, bringing the power of thousands and thousands of volunteers
from all around the country to adjudicate according to "reasonableness"
the many individual disputes not warranting the attention of the few
paid workers, a court or a lone "ombudsman".
[Some further remarks are in the next issue, TE News 179, and in
defience of chronology I am adding this link here.]
Scattered
Thots
* For a number of years, farmers have been going out of business in
droves. Rising costs for everything, poor weather and now scarcity and
cost of fertilizers has reduced crop yields and now rising interest
rates on their large loans have tipped more of the remainder over the
edge. Billionaires like Bill Gates buy up the land cheap, hundreds or
thousands of farms. They're "just an investment" - they have no
intention of farming. So there are no farms to be had by erstwhile new
farmers, and a staggering amount of producing acreage has been and is
being lost.
And the reduced yields are of course affecting those
farmers that remain. As food costs rise (50% food inflation over 3
years according to Neil-McCoy-Ward on youtube), the Canadian government
has decided hand out extra money to those on pensions and assistance to
help cover the increasing costs.
What I want to know is how this helps farmers to stay in
business and to increase the food supply? Prices can only continue to
rise rapidly if there isn't enough to go around. Ah well, all the
disastrous bad decisions by those of influence are doubtless all part
of the "Great Reset" or Christ Michael's "Correcting Time" plan or
whatever it is you want to call what is rapidly coming to a head like
an oncoming derailed train.
* We Discovered Why Trains Keep Derailing
https://www.youtube.com/watch?v=olnVwQzQZRs&ab_channel=MorePerfectUnion
-- Now 1700 derailments per year. It turns out it's the wheel bearings.
When the grease dries up the wheels melt off. Of course there are a few
other factors like the railroads now being owned by "hedge funds" who
have no interest in or knowledge of running railroads, and who have
pared labor and maintenance to the bone for a temporary increase in
profit margin.
-- 31G$ merger tween CANADIAN PACIFIC and Kansas City Southern was just
approved by US REGULATORS! When did CANADIANS approve anything like
this?
-- "Six major railroads left IN AMERICA after mergers over the decades"
One was CN! When did CN become an AMERICAN railroad?!?
* In a mine near the town of Ytterby, Sweden, four elements were first
discovered and all named for the town: Yttrium, Ytterbium, Erbium and
Terbium. Someone went to Sweden and while he was there he asked just
how that town name was pronounced: "Ooterboo." (!) This seems to make
little sense except that in Cryllic alphabet "Y" is pronounced "oo".
(But the rest of the letters are latin. ? )
This means that two of the elements would be "as written"
but the other two apparently are correctly pronounced "Ootrium" and
"Ooterbium". ? And that's probably how they're pronounced in Sweden.
* People keep "finding" or is it "creating" "new elements". But the
highest atomic numbered element that can be created by neutron
bombardment is 100, Fermium. And the Urantia Book says that if there
are more than 100 electrons orbiting a nucleus, it disrupts virtually
instantaneously. And this element, #100, is the highest numbered
element that was found in the fallout from hydrogen bomb tests, along
with all of the lower numbered elements.
Thus we see that anything beyond Fermium is a highly
artificial, short lived curiosity that never could nor will ever be
found in nature. Anything beyond that doubtless exists only as a
positively charged ion with no more than 100 electrons orbiting it. It
bugs me seeing these silly names of "elements" beyond 100 that can
never be seen or made use of in any way, absurdly "extending" periodic
tables. (Apparently Fermium was named not for Enrico Fermi, but for the
French word "Fermé" - "Closed" as in "case closed, there's no more
elements".)
* Colonel Douglas MacGreggor was saying all fall and winter that he
expected the Russians to make a main assault sweeping through Ukraine
as soon as the ground froze hard so they could roll through the open
fields instead of just along the paved roads. Recall that in WWII the
Germans were stopped in the autumn by the mud in Ukraine. (There
weren't any paved highways in Ukraine back then!) His theory was never
put to the test because it stayed warm and stayed warm -- there was
virtually no winter in Ukraine, and indeed all Europe was asking "What
happened to winter this year?" (That also surely prevented a disaster
with heating fuels. But I digress.)
Now it is expected that they are waiting for the spring
rains to end and the ground to harden for the same purpose, because
still or again it's all mud and that has hampered even local operations
on both sides. My next question is: will it rain all summer now? That
would seem to be the fitting next step to keep this senseless war from
ending.
It has become a grueling and horrendous war. Since autumn
it has become like World War One with trenches and fortifications and
neither side daring or able to advance very far, very fast, even when
it makes some sort of breakthrough, because units on the move are
spotted by observation drones and become artillery targets for mass
annihilation. Weapons of destruction are advancing to the point that
the combatants seldom even see each other. Soldiers are killed at too
great ranges, mostly by artillery including shells, rockets, missiles,
"suicide drones" and now "thermobaric" weapons. Strategy and tactics
are changing rapidly and radicly.
* D. Trump claims if elected president again he would end the Ukraine
war "within 24 hours". I've said before that Biden could do the same
any time he wanted, but I gave him a week. Remember how after the 2014
US sponsored coup in Kiev the BBC and other journalists were so
concerned about the rise of neo-nazism in Ukraine? Then remember how
the issue died out of view for four years? The shelling of civilians in
Donetsk petered out. It all seemed to go into low gear as the
military-industrial-congressional-banking oligarchy in Washington had
to work behind Trump's back, because he surely would never have
approved of what was being done.
Now with this "threat" to end the war and make vital
reforms ringing in their ears, fearing he might regain the office, they
presume to arrest him on what are at most minor misdemeanors that (even
if true and if the statute of limitations has not long since expired on
them) might merit a fine or two -- and that no other "honest Abe",
"clean as a whistle" political figure seems to have ever been penalized
for.
* As long as people with hearts full of greed and hatred are in charge
including behind the scenes in unelected positions of influence - and
such types have now managed to expel nearly everyone else - our
societies will continue to deteriorate into chaos. National and
regional governments will finally fail and as I've said before,
civilization will have to be rebuilt from the family and community
level up.
* We could go far beyond just "ending the war". If it were possible I
would prefer to see the Ukraine issue settled the democratic way. Each
oblast (or perhaps regions broken up even smaller) would be offered
something like the following choices, in a manner not so dissimilar to
parts of Europe after World War One:
1. Be part of Ukraine
2. Be part of a new "Western-Rus" country (by whatever name it might
pick)
3. Become part of Russia
4. Become part of Belarus
5. Become part of Poland
There might even be a few more choices - Hungary and
Bulgaria (IIRC) as well as Poland (Lvov region) and Russia (at least
Donbass, Crimea) lost lands to Ukraine by decree under Stalin and
Khrushchev. And the boundaries of the plebiscite might extend somewhat
beyond Ukraine's present borders in case some nearby might want to join
Ukraine or one of the other options. (East of the Dniester in Moldova
("Transnistria") where they speak Russian might like the "Western-Rus"
option... and why does poor Moldova have no access to the Black Sea, so
near? Can we not fix that at the same time?) And if Lvov might want to
rejoin Poland, perhaps the part of Germany given to Poland should also
have a vote as to which administration they would prefer to be under?
Voting would of course be by choice ranking so that voting
for what one wants most doesn't prejudice one's chance of getting what
one wants second most or even third. Obviously each region would be
interested in specific choices and not in all of them. Fine! This is
the voting system that will work in all cases.
Wouldn't that produce the best outcome - the bulk of
everyone getting what they most - or at least second most - want? No
fuss. No war.
There may be many who would prefer the "Western-Rus"
option. We will probably never know. Presently the regions under
Russian control were only offered whether to join Russia or not, under
conditions where they desperately need Russia's military help and while
Russia has a brilliant and well liked leader who will be gone some day
not so far off. So it's no surprise they voted over 90% "Da!" (Two
choices is of course far better than no choice at all - as is presently
the case in all the regions under Kievan control!)
Unfortunately there's far too much politics and too many
politicians with too many agendas, with too little imagination and too
few ideals to allow adoption of such a satisfactory, democratic
solution under present conditions. So we have war instead.
* I thought we in the West didn't like Lenin, Stalin or Khrushchev? Yet
we want to restore Ukraine's 1991 boundaries, the very boundaries those
despots created by transferring more and more bordering regions into
Ukraine? Why do we want to ratify and approve of Lenin, Stalin and
Khrushchev creating this polyglot region which subsequently became a
"country"?
* Putin has canceled 20 billion US$ of debt owed to Russia by African
countries. These debts were mostly accumulated during the Soviet era by
countries espousing the USSR's communist philosophy. Doubtless some of
the original debt has been long since repayed, and one wonders how much
of this remainder could ever have been when nations across central
Africa are now starving. Still it is a magnanimous gesture. The USA
never forgave Britain its World War Two debt. IIRC the last payment was
finally made well after Y2K.
* I had found that my cell phone was making my leg vibrate and pulsate
right where it sat in my pocket. I stopped carrying it and the problems
gradually went away. Then I thought some trouble with my right leg must
be from my car key fob, and I stopped carrying that too. But nothing
changed. Now I think the key fob had nothing to do with my right leg
troubles. It is surely far lower power than the cell phone, and
hopefully not transmitting very much.
(I think it's little mites coming off the firewood and
getting into my pores in my thigh. I've been wearing a glove now to
keep them off my hands, and killing them on my leg with a drop of witch
hazel, applied and wiped off after a moment. But they keep coming back.
I got a whole bunch again recently, all at once. I was perplexed and
then realized it was from the sawdust (complete with mites) coming off
the chainsaw and hitting my right leg when cutting firewood! I'll wear
rain pants or something else impervious next time. Is it only me they
manage to infest?)
ESD
(Eccentric Silliness Department)
* Lightning never strikes the same shrub twice.
* Master mathematician Jesus warned us that "Greed is the square root
of all evil."
* Military spokesman says "enemy underground stronghold must be raised
to the ground."
* "There are consequences for abolishing consequences." - "Uncle Frank"
(Youtube video comment)
"in depth reports" for each project are below. I
hope they may be useful to anyone who wants to get into a similar
project, to glean ideas for how something might be done, as well as
things that might have been tried, or just thought of and not tried...
and even of how not to do something - why it didn't work or
proved impractical. Sometimes they set out inventive thoughts almost as
they occur - and are the actual organization and elaboration in writing
of those thoughts. They are thus partly a diary and are not extensively
proof-read for literary perfection, consistency, completeness and
elimination of duplications before publication. I hope they may add to
the body of wisdom for other researchers and developers to help them
find more productive paths and avoid potential pitfalls and dead ends.
Electric
Transport
Magnetic Variable Torque Converter with Planetary Gear
One day I drove the truck
out of the garage to show it to someone. Well out of the garage,
something went clunk and it was slipping. Murphy's law in action! We
pushed it back in.
But this gave me an idea: I could cut a short keyslot into
the socket of the driveshaft - not the usual "drifting" (is that the
right name?) for which I had no tool, but carefully cutting right
through one wall with the angle grinder, making a key to fit, and
putting a pipe clamp (or something much heavier) over the whole thing.
It would be much harder for it to break loose and slip if there was a
shaft key in it. (I am simply assuming that's the join that's slipping.
could be rong.)
[10th] I was rong. It was the alume rotor again. (I'll do the above
shaft key anyway.) The SDS hub just wasn't tightening down on the
shaft. I think I'll cut deeper grooves into it so it reduces diameter
more easily. Also I'm looking for a thicker and slightly bigger chunk
of pure alume to make a new rotor with stronger magnetic interaction,
so I'll be changing that whole little assembly. (Somehow I have a
feeling there's a piece or two at the refuse transfer station, but the
snow has kept me from being able to see much there. I keep thinking
it'll be gone in a day or two like usual, but no luck for weeks so far.
When some melts, it soon snows again.)
[21st] I contacted a metal company in Winnipeg about getting 1/2 inch
pure alume for the disks, including looking forward to doing a 12.5
inch one for the Sprint (or the Echo?). I asked for 10.5 inches, since
the magnetic field of the 10 inch magnet rotor doesn't stop abruptly at
its outer edge. The web site said they had "1100 alloy" (pure alume)...
but they didn't.
[29th] I tried a couple more places over some days. I knew pure alume
isn't as common as alloy, but I didn't know it was virtually impossible
to get. Metal Supermarket said I would have to buy a whole sheet (5
feet by 10, was it?); they wouldn't cut it. Just the shipping would be
horrendous, never mind the price of the whole sheet. Only one place
offered me a cut piece: 1100 $ for 2x2 feet, 1/4 inch thick! (I suspect
that might be the same price as a whole sheet: they would cut it and
keep the rest!) Did I have to melt down pop and beer cans? But even if
I did, I couldn't cast the whole rotor in one melt with my small
crucibles, and pieces melted separately absolutely haven't the
slightest tendency to stick together.
Later someone mentioned that only the main part of the can
was pure alume. The top and the tab are alloy. In that case one would
have to melt down an awful lot of cans to get a kilogram for a rotor.
Cast Copper Rotor?
At that point I started thinking buying copper would be
cheaper - or at least, available - as well as being even more
conductive. (Something like 401 versus 237 for pure alume versus 167
for alume alloy). If I used copper I could go thinner. 1/4 inch or 5/16
inch copper, closest to the magnet rotor, would probably work about as
well as 1/2 inch+ of pure alume. I could even cast just a ring of
copper (ie, with no center) to interface with the magnets, and bolt it
to the present rotor?
This day I visited Steve. I lucked out. He had 5(?)
containers full of scrap copper bits, which were sitting on the front
steps at that moment because he was moving from his house (he sold it)
to a nearby one (free rent for fixing it up). Come to think of it, I
have a few scraps myself, but he had a lot weight of copper. He wasn't
sure what he was going to do with them. And I had that new furnace for
melting metals including copper. So now I thought of casting it instead
of buying a piece and having it shipped here. Maybe (unlike alume) if I
cast copper in two or more pieces they would fuse together? It was
still likely to be a couple of hundred dollars or more to buy a 10.5
inch square piece, 1/4 inch thick.
He said to take a couple of the containers. I got them
home and weighed them 31 pounds - 14 Kg! That should be way more than
enough, which would be 2-3 Kg... perhaps castable in one melt, tho with
a very full crucible. A larger (12.5 inch) one for the Sprint might be
5 or 6 Kg, so it would do me for that potential project as well. Also
at the present spot price of copper, that's over 150 $ worth, which
I'll pay him. Plenty potentially for both rotors, and even at that
price much the cheapest idea yet.
And copper should give the optimum result. Why would I
settle for anything less, anyway? It's easy to separate the rotors a
bit if the interaction is too strong. "Too strong" is the opposite of
the problem so far.
I think I need some more molding plaster and silica to
make a smooth high temperature mold. I've had some in a bag for years,
but looking at it, it's only enough for a piece of jewelery or
something. Or maybe I could use sand and just use the plaster/silica
mix to make the inner face smooth? So... looks like a mold box, and a
model disk/ring piece from (?)plywood must be next.
Hmm... the thin but tall graphite crucible that came with
the little furnace holds 1Kg of copper pieces. The ceramic cup crucible
that I thought was bigger holds 750 grams. So much for perceptions!
Someone who's done it before recommended I not try casting
copper. I plan to anyway. Outside of course. Safety gear. I do note
that I should be blowing the air away and not breathe any. I think the
electric furnace will offgas much less than the oxyacetylene torches
I've seen people using to melt copper on youtube. (And they were
melting only tens or 100 grams or so. Melting a Kg would use up a lot
of gas!)
One other consideration he mentioned was that melted
copper can turn to "jello" and not pour. In that case zinc had to be
added. But these scraps are about 3% zinc already. I trust that's not
enough zinc to seriously reduce the conductivity but enough to solve
the potential problem.
Other "Green" & Electric Equipment Projects
Open Loop Air Heat Pumping
[24th] I saw the "Lontra" air
compressor, essentially a "bicycle tire pump" bent around into a circle
and always pumping around that circle. It was said to be 20 or 30 %
more efficient than any other large air compressor. And hence probably
30 or 40 or 50 % more efficient than a small compressor. This would
start to bring to life that promise for a heating COP of 10 or more
that the technology (including the passive outdoor heat exchanger)
seems to hold.
How could it be made more simply? Something I might manage
to do. On this morning an answer came to me. The circular piston
assembly could be an inside [or outside] ring gear, with the teeth of
the gear even with the inside edge of the piston so as to span 360°
without interruption. At the point in the outer pipe where air is
brought in anyway, a gear would go through the inner side to mesh with
the ring. Bearings on the piston ring assembly or inside the pipe would
guide the assembly so it never rubbed on a stationary wall, instead
"floating" on the bearings. The end valve that blocks the circle pipe
while the air is compressing would have to be double and fit around the
piston gear assembly. (Dang, a leaky spot!) The two pieces would
retract to the top and bottom as they were hit by the piston itself so
it could pass to begin the next stroke. (Of course there would be a
one-way flap or valve to prevent the compressed air, once having left
the circular pipe, from returning.)
How to get the piston ring assembly into the pipe? On the
face of it, this looks virtually impossible. It would seem that the
pipe will have to be made as a top and a bottom side that bolt
together. Materials: alume pipe and UHMW-PE piston assembly? Rats, I
have an entirely too coarse set of gear cutters. I'll need to order one
that cuts much finer teeth. (And how will I hold the ring to cut the
gears, unless it is very small? Well, maybe it doesn't need to be
anything like as big as I've been thinking? Maybe I'll keep it under 10
inches diameter so it fits on my lathe, too.) And in fact, maybe it
doesn't need to be gears... maybe for a small unit it could just be a
rubber(?) cylinder rolling against the side fo the ring? Bonus: if the
pressure got too high owing to some blockage, the cylinder and ring
would slip. (Is that a bonus or just a way to fast wear-out?)
...Or maybe an actual simple "bicycle type" pump with a
long, narrow tube with a low mass piston and reciprocating parts, would
also be quite efficient? The worst inefficiency in a bicycle pump is
the skinny hose the compressed air has to go out through. That can
easily be changed! It only needs to make 50 or 60 PSI, so it doesn't
need to be as robustly built as a higher pressure one. I'm sure it
would be much the easiest to make, which could finally get the project
moving along. I'm now leaning to this. But I'm still wondering for it
how to employ the cooled, exhausting, but still compressed air to help
pump the new air, which as I recall saves 29% of the energy.
Electricity Storage
New Chemistry Batteries
At the end of February I
had wrecked my nickel mix by accidently soaking it in toluene instead
of acetone. I figured it probably wouldn't work well after that and
discarded it.
So instead I put together a revised manganese-zinc cell
about the start of the month. I used wax to "pot" it, but this wasn't
very successful as it still leaked even after taking a heat gun to it
and remelting it.
 I put in a copper mesh current
collector and painted it
I put in a copper mesh current
collector and painted it
with zirconium silicate to raise the hydrogen overvoltage.
It didn't seem to discharge itself between electrodes, but
it was somewhat short on MnO2. I had simply used all the dry cell
MnO2+graphite (+Sm2O3) that was left over, a few grams. The voltage
dropped much faster in a discharge test than the previous cell, and it
only put out one amp. Charging current also seemed to be halfed. It did
gradually charge up to 1.5 volts, but it took a couple of days and
after half a day it was only drawing 12mA.
Subsequent tests showed continually reducing capacity, so
maybe trying to recharge to MnO2 just wasn't working well. Nobody else
has got it to work, so should I be surprised? But I also consider that
the fine dry cell nano-powder isn't the best for all the particles. I
found some coarser "granular" manganese dioxide in my stash. I also
looked at Mn metal powder and regular MnO2 powder under the microscope
- three quite different shapes and sizes. Variety might help? What else
could I try? What would be a good additive or process? Monel powder?
[7th] It seemed to stabilize... at the unsatisfactory low level, but it
wasn't getting worse. It might have to do with the cell leaking, too,
so I should try again to seal it up. Instead I decided to redo the MnO2
electrode and see if I could get better performance.
[9th] I took the cell apart by melting the wax with a hot air gun (the
big advantage of wax over epoxy).
Neither the MnO2 powder nor the zinc came cleanly off the
separator paper so it ripped up and I redid it. I cut 5 new separator
papers from a sheet of watercolor paper and soaked them with methyl
benzene (toluene, toluol) twice, the second time being just as they
were about dry from the first time.
When the toluene was dry I painted them with sodium
dodecylbenzene sulfonate. At first I had the SDBS mixed quite thickly,
but it seemed to me it wasn't penetrating much into the paper, so I
thinned it with more water to be sure it would soak in.
I changed the manganese electrode completely. Instead of
dry cell MnO2 I used the somewhat "granular" MnO2 from the pottery
shop. Instead of graphite to raise conductivity I tried monel powder
(from AEE, <325 mesh). And I added some Sm2O3 to raise the oxygen
overvoltage (not sure that's necessary or useful with MnO2 as its
reaction voltage is lower than NiOOH):
MnO2 - 44.8 g - 88%
Monel powder - 5.8 g - 10%
Sm2O3 - 1.0 g - 2%
----------------
'50 g' (51.6 g)
I mixed these in a beaker, then I poured in acetone until it was over
the top even after I mixed it in and all the powder was soaked - a
little deeper than I had meant - and left it overnight to evaporate.
(No more melting PVB battery cases with remnant acetone!)
[10th] Overnight wasn't long enough. It's interesting that the height
of the mix in the beaker is considerably lower than the original dry
powders. It has been considerably "compacted" without even any
pressure, which surely has good prospects for being more conductive.
It finally occurred to me to spread the powder out on the
metal current collector sheet to finish evaporating the acetone, which
I could still smell in the beaker, albeit faintly now. I used 24.15
grams - half of it. Theoretical for MnO2 is 309 AH/Kg. 88% of that is
272. If it gave an actual figure of 200 that would be almost 5
amp-hours worth, with the cell being 1.5 volts. And theoreticly, if one
allowed it to become Mn3O4 at .3 volts less, there's another 2.5
amp-hours at 1.2 volts to be had on top of that - and yet another 2.5
AH at 1.0V for Mn(OH)2 - before the battery actually "dies" entirely,
assuming there is 10 amp-hours worth of zinc. And assuming the Mn_ can
be recharged properly to MnO2 from each of these states.
I did my best to seal the edges of the cell by melting wax
with the hot air gun. It leaked anyway. I set it on its back with the
top slightly raised. That way it's a "dish" with sides.
I filled it and the voltage gradually rose to .90V. I put
it on charge @ 1.6V through .1 ohm. It seemed to take charge much
better than most previous cells. The acetone soak is obviously very
helpful:
~17:30 ~260 mA
17:35 203 mA
17:46 178
18:00 161
(Of course one may ask why it would need charging if it starts out with
MnO2 and zinc metal. I don't have an answer, but my cells always seem
to need initial charging to attain their expected voltage. Perhaps
there's enough air in the cell to oxidize the zinc when electrolyte is
added?)
At this point it supplied a momentary 'short circuit' (.1 Ω) with 1.67
amps. A 10 Ω load brought it down by 100mV from 1.3 to 1.2V. Promising
performance.
18:05 160 mA
18:42 115
Performance is still good. With a 30Ω load, the voltage sits at a
stable point for at least instead of dropping by the second. I'll
hazard a guess that the manganese oxides are charging just fine at pH11
or 12 instead of 14 - at least with the acetone treatment. Has no one
else tried this pH range before? As I've said, everyone seems stuck on
'totally caustic alkali' pH14. And of course metals do different things
at different pH'es. ...or is it the acetone?
20:32 61 mA
24:32 36 mA
 [12th] Well, repeated tests
showed repeating deterioration - lower voltages each time and lower
charging currents after each test. I guess Mn2O3 really doesn't
recharge to MnO2 after all, regardless of the acetone treatment. Nor
does Mn3O4 seem to recharge to Mn2O3. I don't care to know if Mn(OH)2
recharges to Mn3O4 because the cell voltage would be too low to
consider - around 1/2 a volt. Well, it had seemed to be worth a try.
[12th] Well, repeated tests
showed repeating deterioration - lower voltages each time and lower
charging currents after each test. I guess Mn2O3 really doesn't
recharge to MnO2 after all, regardless of the acetone treatment. Nor
does Mn3O4 seem to recharge to Mn2O3. I don't care to know if Mn(OH)2
recharges to Mn3O4 because the cell voltage would be too low to
consider - around 1/2 a volt. Well, it had seemed to be worth a try.
New Nickel Oxides Electrode
 Inner and outer face of a used
cupro-nickel current collector.
Inner and outer face of a used
cupro-nickel current collector.
The outside seems essentially untouched. The side against the electrode
has patches that look like orange copper and others that look like
silvery nickel.
Anyway it's reusable when cleaned off!

Back to nickel. I opened the cell and separated the "+" side. The Mn
oxides were pretty stuck to the separator paper and I hope the toluene
is doing it job of keeping them from getting through to the other side.
But scraping them off the cupro-nickel current collector was where it
was pretty tough going. It was obviously well bonded and thus had a
very good connection. After cleaning it the current collector had the
usual mottled colors of nickel and zinc in patches on the electrode
side, and still seemed quite solid. This still surprises me, knowing
the either copper or nickel alone would have oxidized away.
The zinc sheets however separated from the separator paper
pretty easily - mostly fell right off. Zinc ions didn't seem to be
penetrating it. Yay!
I put a new (treated) separator paper on top of the zinc.
Then I mixed a new batch of nickel oxides. I used the more compact NiO
rather than Ni(OH)2. Any that is exposed to the electrolyte will turn
into hydroxide, but if there's any that's interior and doesn't get wet
it should stay in the more compact & conductive NiO form. I figure
it'll compact better and stay more compacted. And I wonder why others
start off with the hydrated form Ni(OH)2 rather than NiO. Maybe it's
because they make it in solution from NiSO4? But all you need to do is
heat it to over 300°C to convert it to NiO. I suppose it probably
relates to how it performs at pH14. I had 62.8 grams of NiO so I mixed
half of it and kept the same proportions as last time, saving the other
half in case I wanted to try a different mix later:
31.1g NiO - 88%
4.4g Monel - 10%
0.8g Sm2O3 - 2%
-----
36.3
 Then I soaked
it in acetone and stirred it. It went from lumpy to creamy smooth. I
left it overnight.
Then I soaked
it in acetone and stirred it. It went from lumpy to creamy smooth. I
left it overnight.
[13th] It still smelled of acetone but was again a dry powder. But dust
didn't drift out into the air every time it was disturbed like before.
Also I didn't notice a shrinkage like with the Mn oxide. I repainted
the current collector with CaO and put 22.1 grams of the mix into the
cell. 36.3 - 22.1 = 14.2 grams. It looked like there was too much left
over so I weighed it. 27.8 grams! Apparently a considerable amount of
the acetone's substance got incorporated into the powder. Or hadn't yet
evaporated when I weighed it.
27.8 - 14.2 = 13.6 additional grams. So about 6 would have been in the
22 grams that went into the cell, so the cell had about 16 grams of the
dry mix ingredients. 16 x 88% = 14 grams of actual NiO, which by
hydrating would convert to 17 grams of Ni(OH)2. If it would make the
theoretical 289 AH/Kg, that would be 5.0 AH. That was certainly all the
powder that would fit into the cell, maybe 3+ mm thick before
compacting it.
The Ovonics patent for
getting more out of nickel oxides spoke of mixing 3 or more metals 'at
the molecular level' to get higher oxides and higher capacity, but it
was mostly Ni(OH)2. I guess I have 4: NiO, Copper from the monel
powder, samarium oxide, and the calcium oxide painted onto the current
collector. But no cobalt or zinc oxides, and incorporated in a
completely different way with the acetone. How 'molecular' it is would
be an uneducated guess by me. But anyway, testing shows a level of
improvement in my cells that can hardly be attributed to anything else
but the acetone.
Copper Mesh, Cupro-Nickel Mesh?
I was waiting for inspiration on copper mesh current
collectors. I finally had one. Never mind trying to make something from
copper wire: go on line! (Duh!) Aliexpress had a few types and I
ordered about 10 square feet from one very economical source. Then I
looked for cupro-nickel mesh for the positive side. Nothing at
Aliexpress, but there was a company in India [GanpatInd.com] offering
various types. It was "inquire" rather than "shopping cart", but it
looked like they might be accommodating - maybe even make up whatever
you ask for. Cupro-nickel 70:30, ~20 mesh per inch, ~.5mm wires. Ship
to BC, Canada. Let's see what they say! [No reply - from two companies.]
---
Back to the program... I put the cell together, filled it
(it leaked a bit - but I expected it to be worse.) and it said only
about .5V. I put it on charge, 2.25V through .1Ω and it started off
just under 600 mA (14:05 PM). In 10 minutes it was up to 640mA -
presumably as Ni(OH)2 charges to NiOOH it becomes more conductive. I
momentarily gave it a .1Ω load. It put out 2.7 amps. At 25 minutes, 3.4
amps. Minimum target is 5 amps (50mA/sq.cm of interface area. Maximum
expectation is 20 amps - 200mA/sq.cm.) If not ideal yet, it's in the
right order of magnitude. It looks like it just might be there once
charged! It now needs some thicker wires and more secure connections
than alligator clips.
Recalling that when I was pressing down on loose cells by
hand to improve conductivity there was a limit to how much was useful,
I loosened off the clamping screws a little. I still am leery of
ripping the separator paper, and besides, a bit more space for
electrolyte within the electrodes might be helpful as long as
conductivity is still good. It didn't seem to hurt the performance.
I suddenly thought of something long forgotten... When I
was trying to make batteries ten years ago, I would put a 3D printed
vertical grille next to the separator paper. Thus the electrodes were
separated not only by the paper but also by a blank space, filled with
electrolyte of course. The original idea was to give gas bubbles a
space to bubble up to the surface. But that was also why powder seeping
through the paper wasn't killing them, at least not immediately, even
tho I hadn't treated the paper with toluene as I'm now doing. It seemed
to solve some problems. If I keep finding my cells shorting out maybe I
should go back to that?
Over the afternoon and evening short tests showed
improving charge retention and performance. Hopefully the above
paragraph is superfluous. But maybe I should change the orientation
from "tall" to "wide" so bubbles don't have so far to go to the top,
and a wider water reservoir.
Charge had dropped to 225mA, but after a load test it hit
790 for a bit.
[14th] Improvement continued. In the morning voltages were getting
higher, charge current was down to 180mA and it put out 4.39 amps into
a .1Ω load - over 43mA/sq.cm. (And probably another .2Ω in the wires,
but the current was measured across the .1Ω resistor.) That's getting
up to the lower edge of "acceptable" for actual use. By early afternoon
it was over 5 amps momentarily - definitely getting there!
[15th] Further improvement - charge retention after a minute is up 10mV
to 1.993 V and so are the 30 Ω load voltages, dipping under 1.8 V only
by 10 minutes of discharge. After 15 minute load tests current into .1
Ω is around 4.3 amps.
In the afternoon I went to do another 30 ohm, 15 minute
load test. Charging current had dropped another 10 mA to 135. What a
change to see gradual reduction continuing instead of it starting to
climb because there was something getting across the separator! But
it's only been two days. It may yet disappoint me.
I went to get coffee, and promptly forgot about it until
it had gone 29 minutes. It was still putting out 1.719 V, and the
millivolts per minute drop seemed to have got very low. In the early
minutes it was a few millivolts above any previous run.
How long could it now run, delivering how many milliamp-
or even amp-hours? Time for a longer discharge test approached. Before
bed I ran a[nother] 1/2 hour 30 Ω load test. Results were identical to
the previous one virtually to the millivolt. Again it was losing under
3 mV/minute by the end. But charge retention after 1 minute off charge
was down a bit from 2.000 V to 1.992, as was the maximum current into
.1 Ω following the load test, from 4.1 to 3.9 amps. Slightly
disturbing. Did that mean it was too soon to start sealing cells in
epoxy... or that leaking was the problem giving varying results, and I should.
[16th] After squirting in a bit more water I did another morning load
test. It retained to 1.999 V after 1 minute, and after that virtually
all the voltages were about 6 to 8 mV higher than the previous night,
1.721 V at the 30 minute mark. Current into .1 Ω was about the same and
so was initial recharging current. So improvement was there but seemed
to be "law of diminishing returns" now, and it was probably time for a
long load test. Things to do... Maybe this evening?
In the evening charge current was down to 120mA. I ran a
load test with 20 Ω instead of 30. It ran for 20 minutes before
dropping below 1.7 V instead of 30. 2-1/2 hours later I did one with 10
Ω. Sourcing 180 mA instead of 60 or 90, the voltages were somewhat
lower and I went for 1.6 V instead of 1.7. After 15 minutes it fell
below 1.6 V and I ended it. It recovered to almost as high a voltage as
the other tests. I'd say performance was about the same, or maybe I
should say "proportional" to the load.
Since the "RS232 Serial to USB Keyboard" adapter part
arrived a week or two ago, maybe I should finish making and programming
a basic version of the battery load cycle tester for running longer
tests.
[17th] In a second 10 Ω load test voltages were 20-25 mV higher, and
the test ran 18 minutes instead of 15 to drop to the same voltage - 20%
longer. The only worrisome counterindication is that the momentary
'short circuit' current into .1 Ω after running the test keeps dropping
bit by bit, now down to 3.5 amps. That's still probably triple previous
cells.
Late I ran a longer 20 Ω load test, for an hour. Toward
the end it was dropping only 2.5 mV/minute and seemed to just be into
its stride. At the 1h:04' mark it was down to 1.577 V and it would have
delivered over 85 mA-H. If that rate continued it could have run
another 30+ minutes down to 1.5 V, and still longer the lower one
wished to run it to - perhaps another hour beyond that from 1.5
to 1.4 V. On most "definitive" capacity tests I've seen they run cells
down to .9 volts, but when they hit a certain point they start dropping
rapidly. If I want to see if it has amp-hours instead of
milliamp-hours, I need to get that automatic load cycler running. What
to do next?...
[18th] I decided to see how long the cell held its charge. It was
quickly apparent that it had developed some resistance path between
electrodes because the charging current was up a bit. And when
disconnected, in one minute it was down to 1.993 V where it had
previously been holding over 2.0 V. In about six hours it was down to
1.718 V. Next question: is it the zinc, the nickel powder, or something
else? Could something have got around the edge of the separator sheet?
I suppose it will get worse, and then I'll have to open the cell and
look.
Or maybe I'll glue in a 3D printed ring of PVB around the
edges above the separator sheet, pressing its edges together to
really ensure nothing can possibly bypass it around the edge, and see
if that cell lasts for weeks and months without acquiring that
seemingly inevitable self discharge that starts to creep into my cells.
Later it finally occurred to me why a leaking cell would
also deteriorate: If there is no leak, the water inside the cell is
essentially stationary. If there is, adding water/electrolyte at the
top that leaks out at the bottom means water is gradually flowing
through the cell, so I would expect that the SDBS is gradually getting
washed out. When it weakens enough zinc dendrites will start crossing
the separator sheet.
So just the fact that the cell is leaking is probably
enough to cause its demise, perhaps within weeks.
Nickel-Manganates Again
In the meantime, I'll assume the problem is solvable.
Having added a bit of bleach to my jar of previously made powder and
let it dry, I poured in acetone and stirred it up. At one point the
spoon "lurched" at a lump and I splattered some on myself and was glad
I had put safety glasses on, as getting acetone in your eye(s) will
blind you. (None actually hit my face. Still!) When enough acetone is
added, the mix loses all its lumps and becomes creamy smooth. My
supposition is that it dissolves the oxides, which then reform into
new, epitaxial crystals as it evaporates. Epitaxial means that instead
of a pure crystal of one substance, the crystal is a hybrid of two or
more metals. In this case of course, especially manganese and nickel
oxides. So instead of having a grain of manganese powder and a grain of
nickel powder, one has a grain containing both, more or less mixed at
the molecular level. And I surmise that's why previously I had
disappointing results with the idea: charging and discharging the
electrode wasn't sufficient to have it form the desired combined
substance. So I'm trying again now that I've discovered the acetone
(methyl-methyl keytone) trick.
 [19th] I redesigned the
case to be short and wide instead of tall and thin. That way bubbles
don't have so far to go to get to the top and come out in the top
reservoir. And acting on the assumption that zinc ions or nickel oxides
powder has been creeping around the separator sheet, I made a thin
frame to insert and maybe glue to the cell edge, over the top edges of
the sheet. That way it will be sandwiched all the way around the edges.
[19th] I redesigned the
case to be short and wide instead of tall and thin. That way bubbles
don't have so far to go to get to the top and come out in the top
reservoir. And acting on the assumption that zinc ions or nickel oxides
powder has been creeping around the separator sheet, I made a thin
frame to insert and maybe glue to the cell edge, over the top edges of
the sheet. That way it will be sandwiched all the way around the edges.
 It took me 4
tries to get it the right size, and just as I finished the last one, it
occurred to me that since the objective was to seal the edges of the
separator sheet, I might be better off if I could run a bead of
beeswax, or maybe heat glue, around the edge of the paper instead. But
how to get a thin bead?
It took me 4
tries to get it the right size, and just as I finished the last one, it
occurred to me that since the objective was to seal the edges of the
separator sheet, I might be better off if I could run a bead of
beeswax, or maybe heat glue, around the edge of the paper instead. But
how to get a thin bead?
[20th] Maybe instead of a heat glue operation I could 3D print a whole
cover with triangular edges? The idea would be that it would push the
powder inward as it was pressed in, and the sharp edges... Oh wait. If
the paper was even slightly smaller than the space the points would
miss it and it could lift up. (It is amazing on taking apart dry cells
to see how carefully everything is sized and shaped so the electrodes
just miss touching each other. 9 volt cells tend to be very
interesting.)
The nickel-zinc cell I made on the 13th continues to
perform about the same, each test being very similar to previous ones.
The self discharge hasn't got worse like it usually does. But it still
leaks. I keep filling it and there's salt all around it. Is there
anything I can do to stop leaks besides potting the whole cell in epoxy?
I've been minimizing the use of the osmium film, painting
it sparingly on one surface of the zinc sheet. But I'm getting a
maximum of around 5 amps from this cell. 20 would be nicer. It occurs
to me that I might get higher currents if I also spread some 'extra'
into the zinc powder I sprinkle onto the top of that sheet.
I had read that PVB plastic (poly vinyl butyrate) couldn't
be solved in acetone or methylene chloride, but that it could in
alcohol. I decided to glue the top cover on. Nope. Isopropyl alcohol?
Nope. Acetone? Yup - glues! Am I remembering wrong what I was rather
surprised to read, or was the author "out to lunch"? (But I think I
read it in more than one place?)
 When I
actually assembled the cell I used heat glue. But the bead of glue was
thick enough that I think the PVB "picture frame" would be better. Next
time.
When I
actually assembled the cell I used heat glue. But the bead of glue was
thick enough that I think the PVB "picture frame" would be better. Next
time.
I also 3D printed a plastic lid to fit better than the
ones I cut from ABS.
With the cell this far along, ready to squeeze thin and
epoxy shut, I didn't finish it in March (and even well into April.)
Video Series
I'm glad I didn't get my planned video series started
until now, because having so recently figured out most of the problems
I was able to speak with much more confidence than a month ago, or even
than a couple of weeks ago, without having to say "Well, here's some
great discoveries and techniques but I really haven't been successful
yet." I'm going to have to retake many of the "hands on" battery making
clips to show the latest things.
In the morning I decided to take a few clips to start my
planned video series Exploring Battery Making with Craig. I
ended up typing a few more pages and taking all the clips. Somehow in
spite of a few distractions during the day I kept up the momentum and
put together the video in the evening. I redid the final rendering
three times (at 15 minutes per) because somehow on just one video clip,
the audio came out garbled. No rhyme nor reason. It kept seeming to be
fixed in the editor, but when finally rendered into a single video file
it was bad again. The "KDenLive" movie editing program must have had
the bad version cached somewhere - even tho I exited and restarted it.
I ended before 4 AM PDT, which is really "only" 2 AM by the sun in
Haida Gwaii. Still a rather insane time to be up until with dawn about
3 hours off.
[21st] I finally removed the bad clip from the video compilation,
renamed the file, and brought it in again. This time it came out right
and I uploaded it to youtube.
Exploring Battery Making with Craig - Intro
https://youtu.be/R0_ZkBFefDk
In the evening I contacted another company in India about
cupro-nickel mesh for the positive current collectors, the first one
having not replied. (Why are they all in India?) This one didn't reply
either.
[24-25th] I continued editing clips for my video series. On the 26th I
uploaded:
Exploring Battery Making #2 - Preparing a Separator Sheet
https://youtu.be/8-MaCfFwvbY
In this I've given away two of my greatest "secrets":
about the toluene for preventing penetration of the paper by
nano-powders, and about the SDBS which I believe is the answer to the
150 year quest for trying to keep zinc ions from killing batteries.
[April 3rd] Some others seem to have solved the zinc problem now too,
using modified chitosan. Their technique is a bit different in that
they bond their substance to the surface of the zinc instead of putting
it in the separator sheet. One suspects they know what they're doing
better than I, and that I should investigate that.
Exploring Battery Making #3 - Getting/Making Sodium
dodecylbenzenesulfonate
https://youtu.be/F_7Vj8SDw0w
This one is about extracting SDBS from Lemon Fresh
Sunlight dishsoap by ersatz "chromatography" in case others find it
as hard to buy as I did.
Exploring Battery Making #4 - A Zinc Electrode
https://youtu.be/JjwtgkQnwzM
Somewhere in that I'll have to dig out my now decade+ old
method for making acetaldehyde using hexavalent chromium to make the
osmium doped film on the zinc. I'm almost out of it anyway so I need to
make more. Of course, one can buy acetaldehyde from a chemical company.)
I had also earlier taken clips of making the powder for a
nickel manganates electrode. I put them together and added another one
at the end about dissolving the powders in acetone to get everything
mixed at the molecular level within epitaxial crystals. So I have the
video. But I haven't proven that I've made ~~NiMn2O4 yet, or that it
works as a rechargeable electrode. I decided to wait for a successful
result before uploading it. That means making another cell.
Then, with the nickel manganates plus side put in and the
cell all ready to close up and epoxy, I cut down the alder trees and
didn't get back to the cell in March. The "formulating a nickel
manganates" video is ready but I want to make sure it actually works
before I publish it, and do the actual electrode assembly video at the
same time.
As I went along I started thinking that there probably
weren't many DIYers who were going to want to tackle all the
intricacies of making batteries. My target audience then is probably
electrochemists who will see value in some of the things I show, and
present or erstwhile battery manufacturers who may want to set up
actual production.
As usual, some industrialist will doubtless make a fortune
for being the first to market. And maybe more than one of my
chemistries will be put to use. The inventor gets nothing for changing
the world, but if I tried to sell the several ideas or a plan to
manufacture them myself for money, no one would pay me any attention. I
still remember getting "We don't fund that sort of thing." from the
government NRC-IRAP technology representative. And being turned down
for the "new battery" competition. A lifetime with several creative
"world's first" or "world's best" accomplishments seems to mean nothing
when one seeks capital. Especially without "PhD" after one's name.
My
Solar
Power
System
I think maybe I like "photo panes" as a (somewhat) short
form for "solar panels", or "panes" for "panels". That would be
somewhat unambiguous as people usually speak of "windows" rather than
"window panes". And they are, mostly, a pane of glass.
The Usual Daily/Monthly/Yearly Log of Solar Power Generated [and
grid power consumed]
(All times are in PST: clock 48 minutes ahead of local sun time, not
PDT which is an hour and 48 minutes ahead. (DC) battery system power
output readings are reset to zero daily (often just for LED lights,
occasionally used with other loads: Chevy Sprint electric car,
inverters in power outages or other 36V loads), while the grid tied
readings are cumulative.)
Daily Figures
Notes: House Main meter (6 digits) accumulates. DC meter now
accumulates until [before] it loses precision (9.999 WH => 0010
KWH), then is reset. House East and Cabin meters (4 digits) are reset
to 0 when they get near 99.99 (which goes to "100.0") - owing to loss
of second decimal precision.
Km = Nissan Leaf electric car drove distance, then car was charged.
New Order of Daily Solar Readings (Beginning May 2022):
Date House, House, House, Cabin => Total KWH Solar [Notable power
Uses; Grid power meter@time] Sky/weather
Main
DC East Cabin
February
28th 4292.58, 9.75, 45.81, 34.26 => 7.75 [5745@18:30] Snow melted
off panels.
March
01st 4294.21, 9.86, 46.09, 35.00 => 2.76 [5781@18:00]
Snow melting in rain. Didn't get carport panels fixed yet.
02d 4296.29, 10.0, 46.94, 36.48 => 4.55 [50Km;
5817@18:30] More snow! Some sun too.
03rd 4296.92, 0.09, 47.32, 36.84 => 1.46 [85Km;
5861@19:00] Still snow on panels.
04th 4303.26, 0.21, 48.70, 41.14 => 12.14 [20Km; 5895@19:00] Snow
musta melted off panes.
05th 4310.38, 0.30, 50.05, 46.08 => 14.40 [5921@19:00] Sun but still
freezing at night - snow is not all gone, crunchy not slushy.
06th 4316.60, 0.38, 51.32, 50.36 => 9.85 [?]
07th 4319.98, 0.47, 52.27, 52.60 => 6.66 [5981@18:30] -3.2°
this morning. PM until evening: More snow!
08bc 4324.53, 0.54, 53.26, 55.08 => 8.09 [55Km;
6015@18:30]
09ab 4332.01, 0.62, 54.64, 60.08 => 13.94 [someone else charged
their car here 2 hrs; 6063@23:30] Sunny, still cold.
10sk 4337.64, 0.70, 55.98, 63.42 => 10.39 [85Km; 6096@18:30]
11mn4338.75, 0.78, 56.12, 63.85 => 1.76 [40Km; 6133@18:30]
Rain, snow, wind, bletch
12on 4341.79, 0.85, 56.69, 65.48 => 5.31 [40Km;
6168@18:30] some sun, then mor snow
13qc 4345.29, 0.93, 57.49, 67.63 => 6.53 [6207@25:30]
mor snow overnite, ice
14ns 4348.54, 1.05, 58.14, 69.34 => 5.73 [6234@18:30]
15nb 4353.89, 1.13, 59.13, 72.30 => 9.38 [20Km;
6260@19:00] snow, or hail, on porch in AM?
16nf 4358.23, 1.24, 60.13, 74.87 => 8.02 [6287@19:00]
Snow is all gone!
17pe 4362.21, 1.32, 61.03, 77.50 => 6.59 [90Km;
6318@19:00]
18th 4363.33, 1.40, 61.14, 77.94 => 1.75 [55Km;
6363@18:30]
19th 4365.88, 1.48, 61.56, 78.93 => 4.04 [6393@19:00]
20th 4369.74, 1.55, 62.29, 80.97 => 6.70 [6417@18:30]
21st 4376.70, 1.63, 63.77, 85.58 => 13.13 [6443@19:00] A genuine
mostly sunny day! Even kind of warm. (8°?) Fixed Echo sticking brake
22d 4382.85, 1.71, 65.57, 89.05 => 11.50 [20Km; 6472@19:00] Resurrected "A" frame on carport roof
w. 2 panes. #3 was broken.

23rd 4387.23, 1.79, 67.82, 91.49 =>
9.15 [6499@19:00] Reinforced "A" frame properly so it can't get
blown over again. (I trust.)
24th 4392.12, 1.87, 71.02, 94.56 => 11.24 [90Km; 6527@19:00]
25th 4397.68, 1.95, 74.16, 3.21 => 11.99 [55Km;
6564@19:00]
26th 4402.89, 2.02, 77.18, 6.13 => 11.22 [6579@19:00]
Too windy to put up 3rd sun pane on carport.
27th 4411.89, 2.13, 82.49, 12.70 => 20.99 [6605@19:00] Genuine
sunshine! (now too busy for 3rd pane)
28th 4420.97, 2.20, 87.75, 18.50 => 20.21 [6628@19:30] MORR
sunshine! (cut down 3 alder trees)
29th 4429.90, 2.28, 93.24, 24.69 => 20.69 [55Km; 6660@19:30] Yay for
sunshine! (splitting firewood) SAY! The Nissan Leaf is down to "11
bars" battery health. (As of when? I don't know but quite recently I'm
sure.)
30th 4434.70, 2.37, 95.55, 27.35 => 9.86 [55Km; 6692@19:00]
Cloudy. Later, drizzle.
31st 4442.07, 2.47, 4.11, 31.78 => 16.01 [90Km; 6726@19:00]
April
1st 4448.62, 2.54, 8.64, 35.60 => 14.97 [55Km;
6758@19:30] Frost overnight, decent - calm*, then snow, sun, snow,
drizzle.
* Put up new 3rd solar pane on
carport roof on this morning opportunity.

2d 4457.97, 2.62, 15.92, 41.57
=> 22.68 [6784@19:30] Sunny but cold. (High 6.5°?)
3rd 4464.26, 2.70, 20.83, 45.45 => 15.16 [6813@19:00]
4th 4467.39, 2.78, 22.26, 47.03 => 6.22 [25Km;
6848@19:00]
5th 4470.00, 2.85, 23.39, 48.36 => 5.14 [60Km;
6874@19:00] Cold, drizzle, strong wind, big waves & misery. Not
working outside.
6th 4473.05, 2.93, 24.82, 50.02 => 6.22 [65Km;
6906@19:30] Drizzle. Wind finally calmed by evening.
7th 4479.48, 2.99, 29.40, 54.06 => 15.11 [90Km; 6931@19:30]
Worthless bitcoin minor OFF, maybe for the summer? (not that it's
anything like warm out!)
8th 4483.25, 3.07, 31.42, 56.16 => 7.97 [6947@19:30]
More wind, clouds & such crap
Chart of daily KWH from solar panels. (Compare MARCH 2023 (left) with February 2023 & with March
2022 - but note number of solar panels.)
Days of
__ KWH
|
March 2023
(15 then 17 solars -
repaired 2 carport
panes on 22nd)
|
February 2023
(18 then 15
solar panels
after huge wind)
|
March 2022
(15 s. panels)
|
0.xx
|
|
|
|
1.xx
|
3
|
4
|
|
2.xx
|
1
|
6
|
1
|
3.xx
|
|
4
|
3
|
4.xx
|
2
|
1
|
2
|
5.xx
|
2
|
4
|
4
|
6.xx
|
4
|
4
|
5
|
7.xx
|
|
2
|
5
|
8.xx
|
2
|
1
|
5
|
9.xx
|
4
|
|
1
|
10.xx
|
1
|
1
|
1
|
11.xx
|
4
|
1
|
1
|
12.xx
|
1
|
|
1
|
13.xx
|
2
|
|
1
|
14.xx
|
1
|
|
|
15.xx
|
|
|
|
16.xx
|
1
|
|
|
17.xx
|
|
|
|
18.xx
|
|
|
|
19.xx
|
|
|
|
20.xx
|
3
|
|
|
Total KWH
for month
|
298.14
|
132.46
|
214.48
|
Km Driven
on Electricity
|
954.9 Km
(140 KWH?)
|
713.1 Km
(~110 KWH?) |
1001.6 Km
(160 KWH?)
|
Things Noted - March 2023
* Daylight rapidly longer and longer as sun crosses equator at spring
equinox.
* Best March for solar ever by a long shot, thanks in no small measure
to 3 sunny days at the end of the month as well as fixing 2 of the 3
carport panels for the last 10 days.
Monthly Summaries: Solar Generated KWH [& Power used from
grid KWH]
As these tables are getting long, I'm not repeating the log of monthly
reports. The reports for the first four full years (March 2019 to
February 2023) may be found in TE
News
#177, February 2023.
2023 - (House roof, lawn + DC + Cabin + Carport, Pole) Solar
Jan KWH: 40.57 + 3.06 + 28.31 + 21.85 = 93.79 Solar [grid: 1163; car
(est): 130]
Feb KWH: 59.19 + 2.70 + 38.10 + 32.47 = 132.46 Solar [grid: 1079; car:
110]
Four years of solar!
Mar KWH: 149.49 + 2.72 + 53.85 + 92.08 = 298.14 Solar [grid: 981; car:
140]
Annual Totals
1. March 2019-Feb. 2020: 2196.15 KWH Solar [used 7927 KWH
from grid]
2. March 2020-Feb. 2021: 2069.82 KWH Solar [used 11294 KWH from grid]
(More electric heat - BR, Trailer & Perry's RV)
3. March 2021-Feb. 2022: 2063.05 KWH Solar [used 10977 KWH from grid]
4a. March 2022-August 2022: in (the best) 6 months, about 2725 KWH
solar - more than in any previous entire year!
4. March2022-Feb. 2023: 3793.37 KWH Solar [used 12038 KWH from grid]
Money Saved or Earned - @ 12¢ [All BC residential elec. rate] ;
@ 50¢ [2018 cost of diesel fuel to BC Hydro] ; @ 1$ per KWH [actual
total cost to BC Hydro in 2022 according to an employee]:
1. 263.42$ ; 1097.58$ ; 2196.15$
2. 248.38$ ; 1034.91$ ; 2069.82$
3. 247.57$ ; 1031.53$ ; 2063.05$
4. 455.204 ; 1896.69$ ; 3793.37$
It can be seen that the benefit to the society as a whole
on Haida Gwaii from solar power installations is much greater than the
cost savings to the individual user of electricity, thanks to the heavy
subsidization of our power owing to the BC government policy of having
the same power rate across the entire province regardless of the cost
of production. And it can be insurance: With some extra equipment and a
battery, sufficient solar can deliver essential power in electrical
outages however long. (Feb 28th 2023: And it's probably well over
1$/KWH by now the way inflation of diesel fuel and other costs is
running.)
https://www.TurquoiseEnergy.com
Haida Gwaii, BC Canada

 There was a
second Renewable Energy Symposium on the 11th &
12th, at the Haida Heritage Museum, following the one in September 2018
in Old Masset centre. I decided to get a table and show mainly the
battery and torque converter work I had been doing, plus the updated
handheld bandsaw mill and some "Plastic Recycling 2.0" molds and
moldings.
There was a
second Renewable Energy Symposium on the 11th &
12th, at the Haida Heritage Museum, following the one in September 2018
in Old Masset centre. I decided to get a table and show mainly the
battery and torque converter work I had been doing, plus the updated
handheld bandsaw mill and some "Plastic Recycling 2.0" molds and
moldings. I did a few
new "sample size" pieces in the kitchen oven the week before to take to
the event, where I discovered, since I could control the temperature,
that 400°F was about right for both PE and PP.) I started typing up
information pages about the battery research on the 5th, even before
finishing the February TE News Report.
I did a few
new "sample size" pieces in the kitchen oven the week before to take to
the event, where I discovered, since I could control the temperature,
that 400°F was about right for both PE and PP.) I started typing up
information pages about the battery research on the 5th, even before
finishing the February TE News Report. I visualize a
piece of gadolinium coated on opposite sides with copper or graphite,
perhaps moving back and forth. On its outward swing it contacts a
graphite coated magnet, which is cooled by a fan. So it heats up, but
its heat is drawn through the copper and or graphite. Then it goes the
other way where it cools as it exits the magnetic field. Here the other
face (or better, somehow the same face) contacts the cooling side
copper and transfers its coolness into that. If instead of a back and
forth "swing" it is traveling say in a circle, the copper/graphite heat
transfer contacts could be rollers - or the Gd could be in the form of
a roller - to avoid friction. Each stage transfers to the next by a
solid copper or graphite bar, hot to cold, hot to cold.
I visualize a
piece of gadolinium coated on opposite sides with copper or graphite,
perhaps moving back and forth. On its outward swing it contacts a
graphite coated magnet, which is cooled by a fan. So it heats up, but
its heat is drawn through the copper and or graphite. Then it goes the
other way where it cools as it exits the magnetic field. Here the other
face (or better, somehow the same face) contacts the cooling side
copper and transfers its coolness into that. If instead of a back and
forth "swing" it is traveling say in a circle, the copper/graphite heat
transfer contacts could be rollers - or the Gd could be in the form of
a roller - to avoid friction. Each stage transfers to the next by a
solid copper or graphite bar, hot to cold, hot to cold. Moving right
along... also on youtube I saw a rather radicly new design of air
compressor, the "Lontra", much more efficient than any previous one. It
had two rotors at 90° to each other. Through most of the cycle the main
rotor acted like a circular bicycle pump, pushing air out the outlet
port. Then it reach the "end" just past the output port, and the gap in
the other rotor, spinning synchronously, reached the gap in the main
one and they passed through each other, virtually but not quite
touching, letting almost no air pass between and closing off the output
port. Meanwhile the whole circle behind the main "piston" had filled
with new air to be compressed on the next cycle. With narrow air gaps
covered by wide pieces for low leakage there were no parts in friction
with each other, and motion was continuous rotation rather than
reciprocating.
Moving right
along... also on youtube I saw a rather radicly new design of air
compressor, the "Lontra", much more efficient than any previous one. It
had two rotors at 90° to each other. Through most of the cycle the main
rotor acted like a circular bicycle pump, pushing air out the outlet
port. Then it reach the "end" just past the output port, and the gap in
the other rotor, spinning synchronously, reached the gap in the main
one and they passed through each other, virtually but not quite
touching, letting almost no air pass between and closing off the output
port. Meanwhile the whole circle behind the main "piston" had filled
with new air to be compressed on the next cycle. With narrow air gaps
covered by wide pieces for low leakage there were no parts in friction
with each other, and motion was continuous rotation rather than
reciprocating.
 Toward the end
of the month I turned the cell design sideways and put some tiny holes
in, to let gas bubbles rise over a short path and into the upper
reservoir, which can hold more liquid.
Toward the end
of the month I turned the cell design sideways and put some tiny holes
in, to let gas bubbles rise over a short path and into the upper
reservoir, which can hold more liquid.
 I started
making a new cell (3D printed front cover) but didn't get it finished
owing to competing attractions. (Just needs compacting + epoxy, and a
new shape of alume clamps.)
I started
making a new cell (3D printed front cover) but didn't get it finished
owing to competing attractions. (Just needs compacting + epoxy, and a
new shape of alume clamps.) In February a
powerful wind with even more powerful gusts blew over my "A" frame of
solar panels on the carport roof. These three panels installed in this
sunny location out of the tree shadows at a good angle (45°) last May
(2022) add more output to the system than any other three panels. After
waiting for better weather I finally got the frame repaired and screwed
back up on the 22nd and 23rd. One panel was broken so I set it up with
two. (I don't think I could have lifted it with all three panels on it!)
In February a
powerful wind with even more powerful gusts blew over my "A" frame of
solar panels on the carport roof. These three panels installed in this
sunny location out of the tree shadows at a good angle (45°) last May
(2022) add more output to the system than any other three panels. After
waiting for better weather I finally got the frame repaired and screwed
back up on the 22nd and 23rd. One panel was broken so I set it up with
two. (I don't think I could have lifted it with all three panels on it!)
 Someone named
Angus showed up here on the 9th in an old Subaru Brat 4 wheel
drive pickup truck he had converted to electric. It had a
Nissan Leaf motor and three big Tesla car batteries behind the seat.
(36KWH? or was it more?) I forget what the motor controller was (a
Tesla model, IIRC), but there wasn't room under the hood for the
regular Leaf controller, and he had got up to 320KW out of the motor
that the Nissan controller normally limits to 80KW!
Someone named
Angus showed up here on the 9th in an old Subaru Brat 4 wheel
drive pickup truck he had converted to electric. It had a
Nissan Leaf motor and three big Tesla car batteries behind the seat.
(36KWH? or was it more?) I forget what the motor controller was (a
Tesla model, IIRC), but there wasn't room under the hood for the
regular Leaf controller, and he had got up to 320KW out of the motor
that the Nissan controller normally limits to 80KW! He had come up from Victoria to
work tree planting, and someone there (Tom!) said he should stop in and
see me.
He had come up from Victoria to
work tree planting, and someone there (Tom!) said he should stop in and
see me.
 Overgrazing
makes deserts. Here, although it's too damp for a desert, my own
chickens illustrated overgrazing when I moved their yard over a little
bit. The green strip didn't last.
Overgrazing
makes deserts. Here, although it's too damp for a desert, my own
chickens illustrated overgrazing when I moved their yard over a little
bit. The green strip didn't last.
 [12th] Well, repeated tests
showed repeating deterioration - lower voltages each time and lower
charging currents after each test. I guess Mn2O3 really doesn't
recharge to MnO2 after all, regardless of the acetone treatment. Nor
does Mn3O4 seem to recharge to Mn2O3. I don't care to know if Mn(OH)2
recharges to Mn3O4 because the cell voltage would be too low to
consider - around 1/2 a volt. Well, it had seemed to be worth a try.
[12th] Well, repeated tests
showed repeating deterioration - lower voltages each time and lower
charging currents after each test. I guess Mn2O3 really doesn't
recharge to MnO2 after all, regardless of the acetone treatment. Nor
does Mn3O4 seem to recharge to Mn2O3. I don't care to know if Mn(OH)2
recharges to Mn3O4 because the cell voltage would be too low to
consider - around 1/2 a volt. Well, it had seemed to be worth a try.

 Then I soaked
it in acetone and stirred it. It went from lumpy to creamy smooth. I
left it overnight.
Then I soaked
it in acetone and stirred it. It went from lumpy to creamy smooth. I
left it overnight. [19th] I redesigned the
case to be short and wide instead of tall and thin. That way bubbles
don't have so far to go to get to the top and come out in the top
reservoir. And acting on the assumption that zinc ions or nickel oxides
powder has been creeping around the separator sheet, I made a thin
frame to insert and maybe glue to the cell edge, over the top edges of
the sheet. That way it will be sandwiched all the way around the edges.
[19th] I redesigned the
case to be short and wide instead of tall and thin. That way bubbles
don't have so far to go to get to the top and come out in the top
reservoir. And acting on the assumption that zinc ions or nickel oxides
powder has been creeping around the separator sheet, I made a thin
frame to insert and maybe glue to the cell edge, over the top edges of
the sheet. That way it will be sandwiched all the way around the edges. It took me 4
tries to get it the right size, and just as I finished the last one, it
occurred to me that since the objective was to seal the edges of the
separator sheet, I might be better off if I could run a bead of
beeswax, or maybe heat glue, around the edge of the paper instead. But
how to get a thin bead?
It took me 4
tries to get it the right size, and just as I finished the last one, it
occurred to me that since the objective was to seal the edges of the
separator sheet, I might be better off if I could run a bead of
beeswax, or maybe heat glue, around the edge of the paper instead. But
how to get a thin bead? When I
actually assembled the cell I used heat glue. But the bead of glue was
thick enough that I think the PVB "picture frame" would be better. Next
time.
When I
actually assembled the cell I used heat glue. But the bead of glue was
thick enough that I think the PVB "picture frame" would be better. Next
time.
